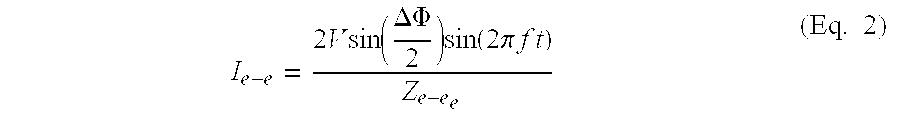US20030195501A1 - RF ablation system and method having automatic temperature control - Google Patents
RF ablation system and method having automatic temperature control Download PDFInfo
- Publication number
- US20030195501A1 US20030195501A1 US10/400,770 US40077003A US2003195501A1 US 20030195501 A1 US20030195501 A1 US 20030195501A1 US 40077003 A US40077003 A US 40077003A US 2003195501 A1 US2003195501 A1 US 2003195501A1
- Authority
- US
- United States
- Prior art keywords
- electrode
- power
- temperature
- electrodes
- tissue
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/04—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating
- A61B18/12—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating by passing a current through the tissue to be heated, e.g. high-frequency current
- A61B18/1206—Generators therefor
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/04—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating
- A61B18/12—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating by passing a current through the tissue to be heated, e.g. high-frequency current
- A61B18/14—Probes or electrodes therefor
- A61B18/1492—Probes or electrodes therefor having a flexible, catheter-like structure, e.g. for heart ablation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00053—Mechanical features of the instrument of device
- A61B2018/0016—Energy applicators arranged in a two- or three dimensional array
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00315—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body for treatment of particular body parts
- A61B2018/00345—Vascular system
- A61B2018/00351—Heart
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00571—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body for achieving a particular surgical effect
- A61B2018/00577—Ablation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00642—Sensing and controlling the application of energy with feedback, i.e. closed loop control
- A61B2018/00654—Sensing and controlling the application of energy with feedback, i.e. closed loop control with individual control of each of a plurality of energy emitting elements
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00696—Controlled or regulated parameters
- A61B2018/00702—Power or energy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00696—Controlled or regulated parameters
- A61B2018/00714—Temperature
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00696—Controlled or regulated parameters
- A61B2018/00726—Duty cycle
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00696—Controlled or regulated parameters
- A61B2018/00738—Depth, e.g. depth of ablation
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00696—Controlled or regulated parameters
- A61B2018/0075—Phase
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00696—Controlled or regulated parameters
- A61B2018/00755—Resistance or impedance
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00773—Sensed parameters
- A61B2018/00791—Temperature
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00773—Sensed parameters
- A61B2018/00791—Temperature
- A61B2018/00797—Temperature measured by multiple temperature sensors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00773—Sensed parameters
- A61B2018/00791—Temperature
- A61B2018/00821—Temperature measured by a thermocouple
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B2018/00636—Sensing and controlling the application of energy
- A61B2018/00773—Sensed parameters
- A61B2018/00875—Resistance or impedance
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/04—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating
- A61B18/12—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating by passing a current through the tissue to be heated, e.g. high-frequency current
- A61B18/1206—Generators therefor
- A61B2018/124—Generators therefor switching the output to different electrodes, e.g. sequentially
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/04—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating
- A61B18/12—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating by passing a current through the tissue to be heated, e.g. high-frequency current
- A61B18/1206—Generators therefor
- A61B2018/1246—Generators therefor characterised by the output polarity
- A61B2018/126—Generators therefor characterised by the output polarity bipolar
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/04—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating
- A61B18/12—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating by passing a current through the tissue to be heated, e.g. high-frequency current
- A61B18/1206—Generators therefor
- A61B2018/1273—Generators therefor including multiple generators in one device
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61B—DIAGNOSIS; SURGERY; IDENTIFICATION
- A61B18/00—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body
- A61B18/04—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating
- A61B18/12—Surgical instruments, devices or methods for transferring non-mechanical forms of energy to or from the body by heating by passing a current through the tissue to be heated, e.g. high-frequency current
- A61B18/14—Probes or electrodes therefor
- A61B2018/1467—Probes or electrodes therefor using more than two electrodes on a single probe
Definitions
- the invention relates generally to an electrophysiological (“EP”) apparatus and method for providing energy to biological tissue, and more particularly, to a radio frequency (“RF”) ablation apparatus for controlling the flow of current through, and the temperature of, a biological site so that the volume of ablation lesions may be controlled.
- EP electrophysiological
- RF radio frequency
- the heart beat in a healthy human is controlled by the sinoatrial node (“S-A node”) located in the wall of the right atrium.
- the S-A node generates electrical signal potentials that are transmitted through pathways of conductive heart tissue in the atrium to the atrioventricular node (“A-V node”) which in turn transmits the electrical signals throughout the ventricle by means of the His and Purkinje conductive tissues.
- A-V node atrioventricular node
- Improper growth of, or damage to, the conductive tissue in the heart can interfere with the passage of regular electrical signals from the S-A and A-V nodes. Electrical signal irregularities resulting from such interference can disturb the normal rhythm of the heart and cause an abnormal rhythmic condition referred to as “cardiac arrhythmia.”
- ablation of the damaged tissue can restore the correct operation of the heart.
- ablation can be performed by percutaneous ablation, a procedure in which a catheter is percutaneously introduced into the patient and directed through an artery to the atrium or ventricle of the heart to perform single or multiple diagnostic, therapeutic, and/or surgical procedures.
- an ablation procedure is used to destroy the tissue causing the arrhythmia in an attempt to remove the electrical signal irregularities or create a conductive tissue block to restore normal heart beat or at least an improved heart beat.
- Successful ablation of the conductive tissue at the arrhythmia initiation site usually terminates the arrhythmia or at least moderates the heart rhythm to acceptable levels.
- a widely accepted treatment for arrhythmia involves the application of RF energy to the conductive tissue.
- AF atrial fibrillation
- Maze procedure a procedure published by Cox et al. and known as the “Maze procedure” involves continuous atrial incisions to prevent atrial reentry and to allow sinus impulses to activate the entire myocardium. While this procedure has been found to be successful, it involves an intensely invasive approach. It is more desirable to accomplish the same result as the Maze procedure by use of a less invasive approach, such as through the use of an appropriate EP catheter system.
- RF energy there are two general methods of applying RF energy to cardiac tissue, unipolar and bipolar.
- a large surface area electrode e.g., a backplate
- the backplate completes an electrical circuit with one or more electrodes that are introduced into the heart, usually via a catheter, and placed in intimate contact with the aberrant conductive tissue.
- electrodes introduced into the heart have different potentials and complete an electrical circuit between themselves.
- the flux traveling between the two electrodes of the catheter enters the tissue to cause ablation.
- the electrodes are placed in intimate contact with the target endocardial tissue.
- RF energy is applied to the electrodes to raise the temperature of the target tissue to a non-viable state.
- the temperature boundary between viable and non-viable tissue is approximately 48° Centigrade.
- Tissue heated to a temperature above 48° C. becomes non-viable and defines the ablation volume.
- the objective is to elevate the tissue temperature, which is generally at 37° C., fairly uniformly to an ablation temperature above 48° C., while keeping both the temperature at the tissue surface and the temperature of the electrode below 100° C.
- a basic configuration of an ablation catheter for applying RF energy includes a distal tip which is fitted with an electrode device.
- the electrode device is the source of an electrical signal that causes heating of the contacting and neighboring tissue.
- the electrode device may include a single electrode used for emitting RF energy. This single electrode acts as one electrical pole. The other electrical pole is formed by the backplate in contact with a patient's external body part.
- a RF source is applied to the electrode.
- the RF source is typically in the 500 kHz region and produces a sinusoidal voltage. When this is delivered between the distal tip of a standard electrode catheter and a backplate, it produces a localized RF heating effect and produces a well defined, deep acute lesion slightly larger than the tip electrode.
- a lesion having a larger surface area than that produced by a single electrode in a unipolar arrangement may be required.
- numerous ablation catheters have been designed.
- an electrode device having four peripheral electrodes which extend from a retracted mode is used. See U.S. Pat. No. 5,500,011 to Desai.
- the four peripheral electrodes and the central electrode form an electrode array that covers a larger surface area of the tissue than a single electrode.
- the five electrodes When used with a conventional RF power source, and in conjunction with a backplate, the five electrodes produce five lesion spots distributed over the area spanned by the electrode array.
- the lesions produced are discontinuous in relation to each other and there are areas between the electrodes that remain unablated.
- This device must be manipulated so that when expanded, all electrodes are in contact with the endocardium.
- An “end on” approach is required such that the end of the catheter, on which all five electrodes are mounted, is in intimate contact with the target tissue.
- an electrode device having a central electrode and a number of peripheral electrodes which also fan out from a retracted mode is used.
- a backplate is not used; instead the central electrode functions as the reference while the peripheral electrodes have multi-phase RF power applied to them.
- This arrangement also requires the same manipulation of the catheter such that an end-on contact is made by the expanded electrodes, as discussed above.
- the mechanical configuration of both of the above-described techniques comprises an expanding approach.
- an electrode device When used for ablation, an electrode device is typically part of a catheter system. Accordingly, it is desirable to minimize the diameter of the electrode device during introduction to and withdrawal from the patient to lessen trauma to the patient. Therefore, electrode devices having peripheral expandable electrodes must be configured so that the peripheral electrodes are expandable to a large size yet are retractable to as small a size as practical. Such requirements pose design and manufacturing difficulties due to the movement of mechanical parts required for proper operation. Further considerations are the undesirable complexity and increased manufacturing cost associated with an expandable a catheter.
- the invention is directed to an apparatus and a method for controlling the application of energy to, and the temperature of, a biological site during ablation.
- the invention relates to an apparatus for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature.
- the apparatus includes a plurality of electrodes positionable proximal the biological tissue and at least one temperature sensing device located on at least one of the electrodes.
- the temperature sensing device provides a temperature signal indicative of the temperature at the interface between the at least one electrode and the tissue.
- the apparatus also includes a generator that operates under the control of a processor to apply power to each of the electrodes.
- the power has an associated phase angle and, within a time duration, a plurality of alternating on periods and off periods, one set of adjacent on and off periods defining a duty cycle.
- the processor is adapted to, during the on period of each duty cycle, set the phase angles of the power provided to each electrode such that at least two of the electrodes are provided power with different phase angles.
- the processor is further adapted to, in response to the temperature signal, determine the temperature at the at least one electrode/tissue interface; compare the interface temperature to the target temperature and adjust the duty cycle of the power provided to the at least one electrode in response to the comparison.
- the processor is adapted to increase or decrease the duty cycle of the power to the at least one electrode when the at least one electrode/tissue interface temperature is less than or greater than the target temperature.
- the processor is adapted to set the power to the at least one electrode to zero when the at least one electrode/tissue interface temperature is a specified first number of degrees greater than the target temperature to thereby allow the electrode to cool off.
- the processor is further adapted to incrementally increase the power to the electrode until the interface temperature is near the target temperature.
- the apparatus further includes a power interruption device connected to the processor. The processor is adapted to control the power interruption device to interrupt power to the at least one electrode when the at least one electrode/tissue interface temperature is a specified second number of degrees greater than the target temperature.
- the invention in another facet, relates to an apparatus for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature.
- the apparatus includes a catheter having a plurality of electrodes at its distal end. The distal end may be positioned so that the electrodes are located proximal the biological tissue.
- the apparatus also includes a plurality of temperature sensing devices. At least one temperature sensing device is located on a select number of the electrodes. These devices provide a temperature signal indicative of the temperature at the interface between the electrode and the tissue.
- the apparatus also includes a generator operating under the control of a processor to apply power to each of the electrodes. The power has an associated phase angle and, within a time duration, a plurality of alternating on periods and off periods.
- One set of adjacent on and off periods define a duty cycle.
- the processor is adapted to, in response to each of the temperature signals, determine the temperature at the electrode/tissue interface and regulate the temperature of a plurality of temperature zones. Each zone includes at least one combination electrode/temperature sensing device. The temperature is regulated by controlling the power to at least one electrode in the zone.
- the processor is further adapted to regulate the power to a plurality of phase groups. Each group includes at least one electrode. The power is regulated by controlling the phase of the power to each electrode in the group such that during the on portion of a duty cycle the phase in one group is different then the phase in another group.
- the invention in another aspect, relates to a method for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature.
- the method comprises positioning a plurality of electrodes proximal the biological tissue. At least one of the electrodes carries a temperature sensing device for providing a temperature signal indicative of the temperature at the interface between the electrode and the tissue.
- the method also includes providing power to each of the electrodes. The power has, within a time duration, a plurality of alternating on periods and off periods, wherein one set of adjacent on and off periods defines a duty cycle.
- Also included in the method is, during the on period of each duty cycle, providing power with different phase angles to at least two of the electrodes; determining the temperature at the at least one electrode/tissue interface in response to the temperature signal; comparing the at least one electrode/tissue interface temperature to the target temperature; and adjusting the duty cycle of the power provided to the at least one electrode in response to the comparison.
- the invention in another aspect, relates to a method for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature.
- the method comprises positioning a catheter having a plurality of electrodes at its distal end so that the electrodes are located proximal the biological tissue.
- a select number of the electrodes carry at least one temperature sensing device.
- the device provides a temperature signal indicative of the temperature at the interface between the electrode and the tissue.
- the method also includes providing power having an associated phase and, within a time duration, a plurality of alternating on periods and off periods, one set of adjacent on and off periods defining a duty cycle.
- the method further includes, in response to the temperature signals, determining the temperature at the electrode/tissue interface; individually regulating the temperature of a plurality of temperature zones, wherein each zone comprises at least one combination electrode/temperature sensing device.
- the temperature is regulated by controlling the power to at least one electrode in the zone.
- the method further includes regulating the power to a plurality of phase groups, wherein each group comprises at least one electrode.
- the power is regulated by controlling the phase of the power to each electrode in the group such that during the on portion of a duty cycle the phase in one group is different then the phase in another group.
- FIG. 1 is a schematic diagram of an ablation apparatus including a power control system, electrode device and backplate;
- FIGS. 2 - 1 and 2 - 2 form a block diagram presenting more detail of a power control system in accordance with aspects of the invention, showing phase angle control, duty cycle control, and impedance and temperature monitoring;
- FIG. 3 is a diagram of a multi-channel ablation apparatus in accordance with aspects of the invention wherein a single microprocessor controls the phase angle and duty cycle of each channel individually;
- FIG. 4 depicts a first power waveform having a first phase angle and alternating instances of peak power and very low power
- FIG. 5 depicts a second power waveform having a second phase angle different from the first phase angle and alternating instances of peak power and very low power;
- FIG. 6 presents a time frame (TF) diagram showing a fifty-percent duty cycle
- FIG. 7A depicts the phase relationship and voltage potential between the first and second power waveforms having first and second phase angles respectively, as a function of time;
- FIG. 7B depicts the phase relationship and voltage potential between the first and second power waveforms having second and first phase angles respectively, as a function of time;
- FIGS. 8A, 8B, 8 C, 8 D, and 8 E are schematic diagrams of an embodiment of a power control system in accordance with aspects of the invention with FIG. 8A showing how FIGS. 8B, 8C, 8 D and 8 E are related;
- FIG. 9A is a three dimensional representation of an ablation apparatus having a linear array of band electrodes in contact with a biological site with a backplate at the opposite side of the biological site, in which the phase angle difference between adjacent electrodes of the linear array is zero degrees;
- FIGS. 9B through 9D depict, along the x, y, and z axes shown, the depth of the lesions formed by the ablation apparatus of FIG. 9A showing that the apparatus acts as a unipolar device with multiple electrodes and the resulting lesions are discontinuous;
- FIG. 10A is a three dimensional representation of an ablation apparatus having a linear array of band electrodes in contact with a biological site with a backplate at the opposite side of the biological site, in which the phase angle difference between adjacent electrodes is 180 degrees;
- FIGS. 10B through 10D depict, along the x, y, and z axes shown, the continuity and depth of a lesion formed by the ablation apparatus of FIG. 10A showing that the apparatus acts as a bipolar device with no significant amount of current flowing to the backplate;
- FIG. 11A is a three dimensional representation of an ablation apparatus having a linear array of band electrodes in contact with a biological site with a backplate at the opposite side of the biological site, in which the phase difference between adjacent electrodes is approximately 90 degrees;
- FIGS. 11B through 11D depict, along the x, y, and z axes shown, the continuity and depth of a lesion formed by the ablation apparatus of FIG. 11A showing the greater depth of lesion resulting from the phase angle difference.
- FIG. 12 presents a block diagram of the current flow among electrodes and the backplate through the biological site for adjacent electrodes having different phase angles
- FIG. 13 presents the same block diagram as FIG. 12 with the phase angles between adjacent electrodes reversed.
- FIGS. 14A through 14D present, along the x, y, and z axes shown, the increased continuity, depth, and uniformity of a lesion formed by the alternating phase apparatus and method shown in previous figures.
- FIG. 1 there is shown an ablation apparatus 10 in accordance with aspects of the present invention.
- the apparatus 10 includes a power control system 12 that provides power or drive 14 to an electrode device 16 .
- the power control system 12 comprises a power generator 18 that may have any number of output channels through which it provides the power 14 .
- the operation of the power generator 18 is controlled by a controller 20 which outputs control signals 21 to the power generator 18 .
- the controller 20 monitors the power 14 provided by the power generator 18 .
- the controller 20 also receives temperature signals 22 from the electrode device 16 . Based on the power 14 and temperature signals 22 the controller 20 adjusts the operation of the power generator 18 .
- a backplate 24 is located proximal to the biological site 26 opposite the site from the electrode device 16 , and is connected by a backplate wire 28 to the power generator 18 .
- the backplate 24 is set at the reference level to the power provided to the electrodes, as discussed in detail below.
- the electrode device 16 is typically part of a steerable EP catheter 30 capable of being percutaneously introduced into a biological site 26 , e.g., the atrium or ventricle of the heart.
- the electrode device 16 is shown in schematic form with the components drawn to more clearly illustrate the relationship between the components and the relationship between the components and the power control system 12 .
- the catheter 30 comprises a distal segment 34 and a handle 31 located outside the patient.
- a preferred embodiment of the electrode device 16 includes twelve band electrodes 32 arranged in a substantially linear array along the distal segment 34 of the catheter 30 .
- the electrode device 16 may include a tip electrode 36 .
- band electrodes 32 are arranged so that there is space 38 between adjacent electrodes.
- the width of the band electrodes 32 is 3 mm and the space 38 between the electrodes is 4 mm.
- the total length of the electrode device 16 is approximately 8 cm.
- the arrangement of the band electrodes 32 is not limited to a linear array and may take the form of other patterns.
- a substantially linear array is preferred for certain therapeutic procedures, such as treatment of atrial fibrillation, in which linear lesions of typically 4 to 8 cm in length are desired.
- a linear array is more easily carried by the catheter 30 and also lessens the size of the catheter.
- the band electrodes 32 are formed of a material having a significantly higher thermal conductivity than that of the biological tissue 26 . Possible materials include silver, copper, gold, chromium, aluminum, molybdenum, tungsten, nickel, platinum, and platinum/10% iridium. Because of the difference in thermal conductivity between the electrodes 32 and the tissue 26 , the electrodes 32 cool off more rapidly in the flowing fluids at the biological site. The power supplied to the electrodes 32 may be adjusted during ablation to allow for the cooling of the electrodes while at the same time allowing for the temperature of the tissue to build up so that ablation results.
- the electrodes 32 are sized so that the surface area available for contact with fluid in the heart, e.g., blood, is sufficient to allow for efficient heat dissipation from the electrodes to the surrounding blood.
- the electrodes 32 are 7 French (2.3 mm in diameter) with a length of 3 mm.
- the thickness of the band electrodes 32 also affects the ability of the electrode to draw thermal energy away from the tissue it contacts.
- the electrodes 32 are kept substantially thin so that the electrodes effectively draw energy away from the tissue without having to unduly increase the outer diameter of the electrode.
- the thickness of the band electrodes is 0.05 to 0.13 mm (0.002 to 0.005 inches).
- each band electrode 32 has a temperature sensor 40 mounted to it.
- Each temperature sensor 40 provides a temperature signal 22 to the controller 20 which is indicative of the temperature of the respective band electrode 32 at that sensor.
- a temperature sensor 40 is mounted on every other band electrode 32 .
- every other electrode has two temperature sensors 40 .
- FIG. 1 which shows an embodiment having one temperature sensor for each electrode, there is shown a single power lead 15 for each electrode 32 to provide power to each electrode for ablation purposes and two temperature leads 23 for each temperature sensor 40 to establish the thermocouple effect.
- the drive wire may comprise one of the thermocouple wires or may comprise a common wire for a plurality of thermocouples mounted on the same electrode.
- the inventors hereby incorporates by reference U.S. Pat. Nos. 6,049,737 and 6,045,550 both of which are assigned to the assignee of the present invention.
- FIGS. 2 - 1 and 2 - 2 a block diagram of an ablation apparatus 10 and method in accordance with aspects of the invention is presented.
- a single channel of the power control system 12 is depicted. This channel controls the application of power to a single electrode 32 .
- a channel may control a plurality or group of electrodes.
- a microprocessor 42 which is part of the controller 20 (FIG. 1), provides a duty cycle control signal 44 to a duty cycle generator (“DCG”) 45 .
- DCG duty cycle generator
- the duty cycle generator 45 receives the control signal 44 by an 8-bit latch 46 .
- the latch 46 provides an 8-bit signal 47 to a duty cycle comparator 48 .
- the comparator 48 compares the 8-bit signal 47 to a count from an 8-bit duty cycle counter 50 and if the count is the same, provides a duty cycle off signal 49 to the duty cycle gate 52 .
- the gate 52 is connected to a frequency source (“FS”) 54 , such as an oscillator that produces 500 kHz.
- FS frequency source
- an 8-bit control has a period or time frame of 0.5 msec.
- the electrode is in the off period only 0.25 msec.
- the period or time frame 78 (FIG. 6) is lengthened by use of a prescalar 56 interposed between the frequency source 54 and the counter 50 .
- the prescalar 56 lengthens the period to 4 msec thus allowing for a 2 msec off period during a fifty-percent duty cycle. This results in a sufficient cooling time for the very thin band electrodes discussed above. Other lengths of the period may be used depending on the circumstances.
- a terminal count detector 58 detects the last count of the period and sends a terminal count signal 59 to the gate 52 which resets the gate for continued output of the frequency source signal. This then begins the on period of the duty cycle and the counter 50 begins its count again.
- the duty cycle is set at fifty percent and the 8-bit latch is accordingly set to 128. In another embodiment, the duty cycle is set at ten percent.
- a programmable logic array (“PLA”) 60 receives phase control signals 61 from the microprocessor 42 and controls the phase of the frequency source 54 accordingly.
- the PLA 60 receives the terminal count signal 59 from the terminal count detector 58 and only permits phase changes after receiving that terminal count signal.
- the output signal from the gate 52 during the on period of the duty cycle is provided to a binary power amplifier (“BPA”) 62 that increases the signal to a higher level, in this case, 24 volts.
- BPA binary power amplifier
- the amplified signals are then filtered with a band pass filter (“BPF”) 64 to convert the somewhat square wave to a sine wave.
- the band pass filter 64 in one embodiment is centered at 500 kHz.
- IOT isolated output transformer
- This signal is then sent to a relay interconnect (“RI”) 67 before it is provided as a power output signal OUTn 14 to an electrode 32 at the biological site to cause ablation.
- the power output signal 14 from the isolated output transformer 66 is monitored in one embodiment to determine the impedance at the electrode 32 .
- a voltage and current monitor (“VCM”) 68 is used.
- the monitor signal 69 is converted to digital form by an A-to-D converter (“ADC”) 70 and provided to the microprocessor 42 .
- ADC A-to-D converter
- some or all of the electrodes 32 may include a temperature sensor 40 (FIG. 1) that provides temperature signals 22 (FIG. 2- 2 ) which are used to determine the temperature at the electrode 32 .
- the power 14 in conjunction with the temperature signals 22 , are used to determine the temperature at the electrode 32 .
- Both the temperature signals 22 and the power 14 pass through a temperature filter (“FL”) 73 before being sent to the microprocessor 42 .
- the temperature filter 73 is contained in a printed circuit board separate from the controller 20 and contains its own processor. In either case, the filter 73 filters out any RF noise present in the power 14 so that the signal may be used for temperature monitoring purposes.
- the microprocessor monitors the power 14 and temperature signals 22 only during the off periods of the power 14 duty cycle. Accordingly, negligible RF noise is present in the power line and filtration is not necessary.
- the microprocessor 42 may alter the duty cycle of the power 14 in response to either or both of the impedance or temperature signals.
- the temperature sensed and/or the determined impedance may be displayed to an operator.
- the operator in response may then manually control the duty cycle or other power parameters such as by rotating a knob mounted on a front panel of an instrument.
- a knob mounted on a front panel of an instrument In the case of a multiple channel instrument and catheter, as discussed below, multiple knobs may be provided in this manual arrangement for control over each channel.
- FIG. 3 a multiple channel ablation apparatus is shown. Although only three complete channels are shown, the apparatus comprises many more as indicated by the successive dots. Those channels are not shown in FIG. 3 to preserve clarity of illustration.
- By providing different voltage levels between two electrodes 32 in an array current flows between those electrodes in a bipolar electrode approach.
- By setting the backplate 24 (FIG. 1) at a voltage level different from at least one of those electrodes 32 , current flows between that electrode and the backplate.
- the current flow through the biological site 26 can be more precisely controlled.
- One technique for setting different voltage levels between the electrodes 32 is to maintain a phase difference between them in an AC approach. By setting the backplate 24 at the reference level, current flows between the electrodes 32 and the backplate.
- the single microprocessor 42 which again is part of the controller 20 (FIG. 1), controls the duty cycle and the phase of each channel individually in this embodiment.
- Each channel shown comprises the same elements and each channel produces its own power output signal 14 (OUT 1 , OUT 2 , through OUTn where “n” is the total number of channels) on respective electrode leads (LEAD 1 , LEAD 2 , through LEAD n where “n” is the total number of leads) to the electrodes 32 .
- This multi-channel approach permits more individual control over each electrode.
- the duty cycle of the power applied to each electrode can be individually controlled.
- One electrode may have a ten percent duty cycle while another has a thirty percent duty cycle.
- the signals as shown in FIGS. 4, 5, and 6 , have alternating instances of peak power i.e., “on” periods 74 , and very low power 76 , i.e., “off” periods.
- the output power 14 is a 500 kHz sine wave.
- the number of cycles of the sine wave contained within one on period 74 has been substantially reduced in the drawing to emphasize the phase difference between the first and second output signals OUT 1 , OUT 2 .
- the voltage of each power signal 14 during an off period 76 is substantially zero and during an on period 74 is approximately 350 volts peak-to-peak.
- the power OUT 1 and OUT 2 also have a variable duty cycle for controlling the length of the on period 74 and the off-period 76 within a time frame 78 (see FIG. 6).
- the duty cycle is the ratio of the length of the on period 74 to the length of the entire time frame 78 .
- the effective power is the peak power times the duty cycle. Thus, a signal having a peak power of 100 watts and a 50% duty cycle has an effective power of 50 watts.
- the two power signals OUT 1 , OUT 2 are phased differently from each other.
- the phase angle of each power signal is set and controlled by the processor 42 and PLA 60 .
- Each power signal OUT 1 and OUT 2 has a respective phase angle and those phase angles differ between the two of them.
- the phase angle difference between the power OUT 1 and OUT 2 produces a voltage potential between the band electrodes 32 (FIG. 1) that receive the power. This voltage potential, in turn, induces current flow between the band electrodes 32 .
- FIGS. 7A and 7B The phase angle relationship of the power and the voltage potential produced as a function of time is shown in FIGS. 7A and 7B.
- V e-e 2 ⁇ V ⁇ ⁇ sin ⁇ ( ⁇ ⁇ ⁇ ⁇ 2 ) ⁇ sin ⁇ ( 2 ⁇ ⁇ ⁇ ⁇ ⁇ f ⁇ ⁇ t ) (Eq. 1)
- V voltage amplitude of power
- FIG. 7A shows first and second power OUT 1 and OUT 2 provided to first and second electrodes respectively having a phase angle difference ⁇ with OUT 1 leading OUT 2 by 132 degrees.
- FIG. 7B shows the same power OUT 1 and OUT 2 but with the phase angles reversed where OUT 2 is now leading OUT 1 by 132 degrees.
- FIGS. 8A through 8E schematic diagrams of an embodiment of the ablation apparatus 10 of FIGS. 2 - 1 and 2 - 2 are presented in FIGS. 8B through 8E while FIG. 8A shows how FIGS. 8B through 8E should be oriented in relation to each other.
- the frequency source 54 provides a signal 80 , typically at 500 kHz with a phase angle controlled by the microprocessor 42 through the PLA 60 , to the duty cycle generator 45 .
- the duty cycle generator 45 modulates the frequency source signal 80 to produce the selected duty cycle in accordance with the duty cycle control signal 44 as previously described.
- the duty cycle generator 45 outputs two signals 82 and 84 to the binary power amplifier 62 .
- a dual MOSFET driver U 2 receives the signals, converts their 5V level to a 12V level, and sends each to a transformer T 2 which transforms the signals into 24 V peak-to-peak power.
- the 24V power is then sent to a multi-state driver 86 which includes a configuration of FETs Q 2 , Q 3 , Q 4 , and Q 5 .
- a conducting state of the driver 86 which is typically the on period 74 of the power, these FETs Q 2 through Q 5 conduct and forward the power to a bandpass filter 64 comprising a series LC network.
- a high-impedance state of the driver 86 which is typically during the off period 76 of the power, the FETs Q 2 through Q 5 are nonconducting and no power is sent to the bandpass filter 64 . Instead the FETs Q 2 through Q 5 present a high impedance load to any signals received through the electrode 32 .
- the load impedance on the FETs Q 2 through Q 5 presented by the circuit following the FETs, the electrode, and the tissue is approximately 150 ⁇ but transformed through the output transformer T 3 , it presents a load impedance to the FETs Q 2 -Q 5 of approximately 0.5 to 1 ⁇ .
- the FETs In the off state, the FETs present an impedance of approximately 250 ⁇ which is large in comparison to the transformed load impedance of approximately 0.5 to 1 ⁇ . Therefore, very little power flows when the FETs are in the off state.
- the bandpass filter 64 operates to shape the output signal provided by the binary amplifier 62 from a square wave to a sinusoidal wave.
- the filtered signal 85 then passes to the isolated output section 66 where it is step-up transformed to 350 volt peak-to-peak sinusoidal power at T 3 .
- the power is then split into two identical power signals OUT 1 A, OUT 1 B and provided to two or more respective band electrodes 32 on the output lines LEAD 1 A, LEAD 1 B.
- the isolated output section 66 also includes relays 88 that may be individually opened to remove the power signals OUT 1 A, OUT 1 B from the electrode leads LEAD 1 A, LEAD 1 B when an alert condition is detected, such as high temperature or high impedance at the respective electrode 32 . As previously mentioned these conditions are determined by the microprocessor 42 which receives signals indicative of the temperature and impedance at each of the band electrodes 32 .
- the power from the isolated output section 66 is monitored and representative signals are supplied to an RF voltage and current monitor 68 where in this case, the voltage and current of each output signal are measured to determine the impedance of the particular channel.
- the measured signals are sent to an A-to-D converter 70 (FIG. 2- 2 ) before being sent to the microprocessor 42 for impedance monitoring. If the impedance is above a threshold level indicative of blood clotting or boiling, the microprocessor 42 sends a signal to the duty cycle generator 45 to reduce or discontinue the duty cycle of the power OUT 1 A, OUT 1 B and thus lower the effective power delivered to the band electrodes 32 .
- the temperature at the electrodes 32 is determined by monitoring the power 14 and temperature signals 22 and measuring the voltage difference between the signals. As previously mentioned, in one embodiment of the invention, these signals pass through a filter 73 (FIG. 2- 2 ) before being sent to the microprocessor 42 . The voltage value is converted to a temperature and if the temperature is above a threshold level the duty cycle of the power 14 is reduced. In the case where a single lead is used to provide a signal which is used to determine the temperature as well as provide power to the electrode 32 , the signal from the lead is received on temperature leads 87 , 89 connected at the output side of the relays 88 .
- the duty cycle of each electrode 32 may be individually controlled by the microprocessor 42 .
- the duty cycle of the output signal may be adjusted. For example, one electrode 32 may have a temperature requiring a duty cycle of ten percent, while another electrode may have a temperature which allows for a fifty percent duty cycle.
- every other electrode 32 has a temperature sensor 40 , the electrodes are grouped in pairs with each electrode in the pair having the same duty cycle.
- the electrode device 16 and the backplate 24 are positioned proximal the biological site 26 undergoing ablation such that the biological site is interposed between the electrode device and the backplate.
- the band electrodes 32 (only one of which is indicated by a numeral 32 for clarity of illustration) of the electrode device 16 each receives power OUT 1 , OUT 2 , OUT 3 , OUT 4 having a phase angle on LEAD 1 through LEAD 4 .
- every other electrode 32 receives the same phase angle. Therefore, the phase angle of electrode A equals the phase angle of electrode C and the phase angle of electrode B equals the phase angle of electrode D.
- the electrodes 32 are formed into a linear array as shown.
- a thermocouple temperature sensor 40 is located at each of the electrodes A, B, C, and D and uses the electrode power lead LEADS 1 through 4 as one of the sensor leads. The sensors 40 provide temperature sensor signals 22 for receipt by the power control system 12 .
- alternate electrodes 32 may be grouped together and each may receive the same power having the same phase angle and duty cycle. Another group or groups of electrodes 32 may be interspaced with the first group such that the electrodes of one group alternate with the electrodes of the other group or groups. Each electrode 32 in a particular group of electrodes has the same phase angle and duty cycle. For example, electrodes A and C may be connected to the same power while interspaced electrodes B and D may be connected to a different power output signal.
- the use of individual power signals also provides the ability to disable any combination of electrodes 32 and thereby effectively change the length of the electrode device 16 .
- an electrode device 16 with twelve electrodes 32 receives twelve power signals from a twelve channel power control system 12 .
- the electrodes 32 are 3 mm in length and are 4 mm apart. Accordingly, by disabling various electrodes, a virtual electrode of any length from 3 mm to 8 cm may be produced by the electrode device 16 .
- the backplate 24 is maintained at the reference voltage level in regard to the voltage level of the power OUT 1 through OUTn.
- phase angle difference is established between adjacent band electrodes.
- This phase angle difference may be adjusted to control the voltage potential between adjacent band electrodes 32 and thus to control the flow of current through the biological site 26 .
- V voltage amplitude of power
- I e-b V ⁇ ⁇ sin ⁇ ⁇ ( 2 ⁇ ⁇ ⁇ ⁇ ⁇ f ⁇ ⁇ t ) Z e - b (Eq. 3)
- V voltage amplitude of power
- FIGS. 9A through 11D illustrate various current flow patterns within a biological site.
- the depths and widths of the lesions depicted in FIGS. 9 through 11 are not necessarily to scale or in scalar proportion to each other but are provided for clarity in discerning the differences between the various power application techniques.
- the phase difference between adjacent electrodes 32 is zero degrees, no current flows between the electrodes in accordance with Eq. 2 above, and the apparatus operates in a unipolar fashion with the current flowing to the backplate 24 as shown in FIGS. 9A through 9D.
- the lesions are discrete.
- the lesions 90 are discontinuous in regard to each other.
- the phase difference between adjacent electrodes 32 is set within the range of a value greater than zero to less than 180 degrees, the current flow varies from a deep, discontinuous unipolar pattern to a more continuous, shallow bipolar pattern.
- the current flows as shown in FIG. 11A.
- FIGS. 11B through 11D The continuity and depth of the lesion 94 is illustrated in FIGS. 11B through 11D.
- adjacent electrodes alternated in phase but were provided with power in groups. Electrodes A and C were provided with power at a first phase angle and electrodes B and D were provided with power at a second phase angle, different from the first.
- the phase angle of the power may be adjusted in order to produce a lesion having different depth and continuity characteristics.
- other elements of the electrode device 16 are considered.
- the width of the band electrodes 32 and the spacing between the electrodes are factors in selecting an optimum phase angle.
- the width of the band electrodes is 3 mm
- the spacing between the electrodes is 4 mm
- the electrodes receive power which establish a phase difference of 132 degrees between adjacent electrodes.
- energy is applied to the biological tissue 26 during the on period of the duty cycle in an alternating unipolar-bipolar manner.
- a voltage potential is established between the electrodes 32 and the backplate 24 .
- current flows through the tissue 26 between the electrodes 32 and the backplate 24 .
- a voltage potential is established between at least two of the electrodes 32 rather than between the electrodes and the backplate 24 .
- current flows through the tissue 26 between the electrodes 32 .
- the voltage difference between the electrodes 32 may be established by providing power with different phase angles to the electrodes as previously mentioned.
- some of the electrodes 32 may be connected to a reference potential while others are maintained at a different voltage level.
- the continuity and depth of the lesion produced may be controlled. For example, operating in the unipolar mode for one-fourth of the on period and in the bipolar mode for three-fourths of the on period produces a lesion having a continuity and depth similar to the lesion 94 illustrated in FIGS. 11B through 11D.
- FIGS. 8 B through and 8 E the following devices are shown: Device Part No. Manufacturer U1 GAL6002B Lattice U2 SN75372 numerous Q1 1RFZ34N numerous Q2, Q3, Q4, Q5 1RFZ44N numerous Q7, Q8, Q9 MPF6601 numerous R3, R5 1 ⁇ numerous T1, T4 CMI-4810 Corona Magnetics, Inc. T2 GFS97-0131-1 GFS Manufacturing T5 CMI-4809 Corona Magnetics, Inc.
- the transformer denoted by “T 3 ” is a 1:12 turns ratio, single turn primary, step up transformer wound on a TDK core PC50EER23Z.
- FIG. 12 presents a block diagram of the current flow among electrodes 32 and the backplate 24 through the biological site 26 for adjacent electrodes having different phase angles where the phase angles of the A and C electrodes lead the phase angles of the B and D electrodes. It has been noted that with the approach shown in FIG. 12, the vector sum of the currents flowing through the site 26 is such that more current flows at one or more electrodes than at others. This is shown figuratively with shorter arrows leading to the backplate from the B and D electrodes. Although the ablation volume is greater than in the prior techniques, the ablation volume appears irregular or nonuniform as shown in FIG. 11D. It is desirable to have a more uniform ablation volume, especially as to depth, so that irregular electrical signals do not pass under the ablation volume at a point having less depth and require a repeat of the ablation procedure.
- FIG. 13 presents the same block diagram as FIG. 12 with the phase angles between adjacent electrodes reversed.
- the phase angles of the power at the B and D electrodes 32 now lead the phase angles of the power at the A and C electrodes 32 .
- the change in current flow due to this opposite phasing is represented figuratively with shorter arrows now at the A and C electrodes thus balancing the current flow pattern of FIG. 12. It has been found that by alternating the phase angles such as shown in FIGS. 12 and 13, a much more uniform current flow and much more uniform ablation volume result.
- a cumulative effect of the current flow causes the tissue between all the band electrodes 32 and the backplate 24 to become ablated, depth-wise through the biological site 26 , at a substantially even rate and thus a lesion having substantially uniform depth is produced.
- FIGS. 14A through 14D where an ablation volume 96 is shown, which has much greater uniformity in shape.
- the ablation lesion 96 has a uniform depth and gives rise to a high level of confidence that the ablation volume created with the ablation apparatus in accordance with the invention will successfully destroy the tissue causing the arrhythmia.
- the phase between the electrodes was alternated as shown in FIGS. 12 and 13 only during the off period of the duty cycle. That is, and with reference to FIG. 6, during the entire on period 74 of the duty cycle of one time frame 78 , the phase angles of the power at the A and C electrodes 32 led the phase angles of the power at the B and D electrodes 32 by 132 degrees. During the following off period 76 of the same time frame 78 , the phase angles of the power to be supplied was changed to be opposite those phase angles used during the on period 74 , in preparation for the next on period.
- phase angles of the power provided to electrodes B and D led the phase angles of the power provided to the A and C electrodes by 132 degrees during that entire on period. During the immediately subsequent off period, the phase angles were again changed so that electrodes A and C would lead electrodes B and D.
- the processor 42 monitors the temperatures at the electrodes/tissue interface and automatically makes power adjustments in view of these temperatures.
- the tissue temperature may range from 50° C. to 65° C.
- the temperature of each electrode/tissue interface is monitored and the power applied to each electrode 32 is individually adjusted to maintain the electrode/tissue interface temperature at or near the target temperature.
- An interface temperature within 5° C. degrees of the target temperature is considered to be “near” the target temperature.
- Adjustments to the power are made by changing the duty cycle of the power being applied to the electrode 32 . When the electrode/tissue interface temperature is less than the target temperature, the duty cycle is increased. Conversely, when the interface temperature is greater than the target temperature, the duty cycle is decreased.
- the processor 42 may institute a “soft cutoff.” During a soft cutoff the processor temporarily sets the power to an electrode 32 to zero by setting the duty cycle of the power such that there is no on portion of the duty cycle. During this continuous off period, the electrode/tissue interface is able to cool off to a temperature less than the temperature that caused the cutoff and usually to a temperature less than the target temperature. Once the interface cools off, the processor 42 incrementally increases the power to the electrode 32 until the interface temperature settles to a temperature near the target temperature. The incremental increase in power may be achieved by programming the processor, using well known proportional integral derivative (PID) algorithms, to increase the duty cycle in either a step like or smooth, continuous manner.
- PID proportional integral derivative
- Conditions which necessitate a soft cutoff may be programmed into the processor 42 .
- Such condition may include an interface temperature that is greater than the target temperature by a first specified number of degrees. For example, if the target temperature is 65° C. and the measured interface temperature is at least 5 degrees greater than 65° C., then the electrode may be subject to a soft cutoff.
- the processor 42 may institute a “hard cutoff.” During a hard cutoff, power to an electrode 32 may be interrupted by opening the relay 88 (FIG. 80) associated with the power output providing the power to the electrode 32 . Conditions which necessitate a hard cutoff may be programmed into the processor 42 . Such conditions may include an interface temperature that is greater than the target temperature by a second specified number of degrees that is greater than the specified number of degrees associated with a soft cutoff. For example, if the target temperature is 65° C. and the measured interface temperature is at least 10 degrees greater than 65° C., then the power to the electrode may be interrupted.
- the electrodes 32 are grouped into electrode zones, with each zone including one or more electrodes.
- the electrodes within a zone are adjacent each other.
- At least one of the electrodes 32 within each zone includes a temperature sensor 40 .
- the processor 42 monitors the interface temperatures of each electrode 32 having a temperature sensor 40 and adjusts the power to each of the electrodes within an electrode zone based on the maximum interface temperature in that zone. Accordingly, if the electrode 32 having the maximum interface temperature provides an interface temperature sufficient to warrant a duty cycle increase, duty cycle decrease, soft cutoff or hard cutoff, each electrode 32 within the zone is subject to the same adjustment.
- controller 20 is shown in FIG. 1 as forming a part of the power control system 12 .
- it may take other forms such as an external processor in a separate computer for example.
- duty cycle control and phase control may be performed by circuits other than those shown here. Accordingly, it is not intended that the invention be limited, except as by the appended claims.
Abstract
A plurality of electrodes are positioned at the distal end of a catheter such that the electrodes may be positioned proximal biological tissue. A select number of the electrodes have a temperature sensing device associated with them for providing a temperature signal indicative of the temperature at the interface between the electrode and the tissue. A generator operates under the control of a processor to apply power to each of the electrodes. The power has an associated phase angle and, within a time duration, a plurality of alternating on periods and off periods, one set of adjacent on and off periods defining a duty cycle. The processor is programmed to determine the temperature at the electrode/tissue interface based on the temperature signals, compare the temperature to a target temperature and to adjust the power to the electrode accordingly. Such adjustments may comprise an increase or decrease in the duty cycle, a setting of the duty cycle to zero followed by an incremental increase in the duty cycle or a power interrupt.
Description
- This is a continuation of application Ser. No. 09/738,032, filed Dec. 13, 2000 which is a continuation-in-part of application Ser. No. 09/501,472, filed Feb. 9, 2000, now U.S. Pat. No. 6,200,314, which is a division of application Ser. No. 09/073,392, filed May, 5, 1998, now U.S. Pat. No. 6,059,778.
- The invention relates generally to an electrophysiological (“EP”) apparatus and method for providing energy to biological tissue, and more particularly, to a radio frequency (“RF”) ablation apparatus for controlling the flow of current through, and the temperature of, a biological site so that the volume of ablation lesions may be controlled.
- The heart beat in a healthy human is controlled by the sinoatrial node (“S-A node”) located in the wall of the right atrium. The S-A node generates electrical signal potentials that are transmitted through pathways of conductive heart tissue in the atrium to the atrioventricular node (“A-V node”) which in turn transmits the electrical signals throughout the ventricle by means of the His and Purkinje conductive tissues. Improper growth of, or damage to, the conductive tissue in the heart can interfere with the passage of regular electrical signals from the S-A and A-V nodes. Electrical signal irregularities resulting from such interference can disturb the normal rhythm of the heart and cause an abnormal rhythmic condition referred to as “cardiac arrhythmia.”
- While there are different treatments for cardiac arrhythmia, including the application of anti-arrhythmia drugs, in many cases ablation of the damaged tissue can restore the correct operation of the heart. Such ablation can be performed by percutaneous ablation, a procedure in which a catheter is percutaneously introduced into the patient and directed through an artery to the atrium or ventricle of the heart to perform single or multiple diagnostic, therapeutic, and/or surgical procedures. In such case, an ablation procedure is used to destroy the tissue causing the arrhythmia in an attempt to remove the electrical signal irregularities or create a conductive tissue block to restore normal heart beat or at least an improved heart beat. Successful ablation of the conductive tissue at the arrhythmia initiation site usually terminates the arrhythmia or at least moderates the heart rhythm to acceptable levels. A widely accepted treatment for arrhythmia involves the application of RF energy to the conductive tissue.
- In the case of atrial fibrillation (“AF”), a procedure published by Cox et al. and known as the “Maze procedure” involves continuous atrial incisions to prevent atrial reentry and to allow sinus impulses to activate the entire myocardium. While this procedure has been found to be successful, it involves an intensely invasive approach. It is more desirable to accomplish the same result as the Maze procedure by use of a less invasive approach, such as through the use of an appropriate EP catheter system.
- There are two general methods of applying RF energy to cardiac tissue, unipolar and bipolar. In the unipolar method a large surface area electrode; e.g., a backplate, is placed on the chest, back or other external location of the patient to serve as a return. The backplate completes an electrical circuit with one or more electrodes that are introduced into the heart, usually via a catheter, and placed in intimate contact with the aberrant conductive tissue. In the bipolar method, electrodes introduced into the heart have different potentials and complete an electrical circuit between themselves. In the bipolar method, the flux traveling between the two electrodes of the catheter enters the tissue to cause ablation.
- During ablation, the electrodes are placed in intimate contact with the target endocardial tissue. RF energy is applied to the electrodes to raise the temperature of the target tissue to a non-viable state. In general, the temperature boundary between viable and non-viable tissue is approximately 48° Centigrade. Tissue heated to a temperature above 48° C. becomes non-viable and defines the ablation volume. The objective is to elevate the tissue temperature, which is generally at 37° C., fairly uniformly to an ablation temperature above 48° C., while keeping both the temperature at the tissue surface and the temperature of the electrode below 100° C.
- A basic configuration of an ablation catheter for applying RF energy includes a distal tip which is fitted with an electrode device. The electrode device is the source of an electrical signal that causes heating of the contacting and neighboring tissue. In the unipolar method, the electrode device may include a single electrode used for emitting RF energy. This single electrode acts as one electrical pole. The other electrical pole is formed by the backplate in contact with a patient's external body part. A RF source is applied to the electrode. The RF source is typically in the 500 kHz region and produces a sinusoidal voltage. When this is delivered between the distal tip of a standard electrode catheter and a backplate, it produces a localized RF heating effect and produces a well defined, deep acute lesion slightly larger than the tip electrode.
- In some procedures a lesion having a larger surface area than that produced by a single electrode in a unipolar arrangement may be required. To this end numerous ablation catheters have been designed. In one catheter designed to provide a larger surface ablation area, an electrode device having four peripheral electrodes which extend from a retracted mode is used. See U.S. Pat. No. 5,500,011 to Desai. When extended, i.e., fanned out, the four peripheral electrodes and the central electrode form an electrode array that covers a larger surface area of the tissue than a single electrode. When used with a conventional RF power source, and in conjunction with a backplate, the five electrodes produce five lesion spots distributed over the area spanned by the electrode array. The lesions produced are discontinuous in relation to each other and there are areas between the electrodes that remain unablated. This device must be manipulated so that when expanded, all electrodes are in contact with the endocardium. An “end on” approach is required such that the end of the catheter, on which all five electrodes are mounted, is in intimate contact with the target tissue.
- In another catheter an electrode device having a central electrode and a number of peripheral electrodes which also fan out from a retracted mode is used. During ablation a backplate is not used; instead the central electrode functions as the reference while the peripheral electrodes have multi-phase RF power applied to them. For example, see U.S. Pat. No. 5,383,917 to Desai et al. While this technique provides a more continuous lesion covering a larger surface area of the tissue, the ablation volume is relatively shallow with a nonuniform depth of the lesion. This arrangement also requires the same manipulation of the catheter such that an end-on contact is made by the expanded electrodes, as discussed above. Lesions having a non-uniform ablation volume are undesirable as the depth at one part of the lesion may not be sufficient to stop the irregular signal pathways. Arrhythmia may reoccur because the irregular signals may pass under such an ablation volume and the procedure must then be repeated to once again attempt to obtain an ablation volume having sufficient depth.
- The mechanical configuration of both of the above-described techniques comprises an expanding approach. When used for ablation, an electrode device is typically part of a catheter system. Accordingly, it is desirable to minimize the diameter of the electrode device during introduction to and withdrawal from the patient to lessen trauma to the patient. Therefore, electrode devices having peripheral expandable electrodes must be configured so that the peripheral electrodes are expandable to a large size yet are retractable to as small a size as practical. Such requirements pose design and manufacturing difficulties due to the movement of mechanical parts required for proper operation. Further considerations are the undesirable complexity and increased manufacturing cost associated with an expandable a catheter.
- Hence, those skilled in the art have recognized a need for a structurally stable invasive ablation apparatus and method that are capable of controlling the flow of current through, and temperature of, a biological site so that lesions with controllable surface and depth characteristics may be produced and the ablation volume thereby controlled. The invention fulfills these needs and others.
- Briefly, and in general terms, the invention is directed to an apparatus and a method for controlling the application of energy to, and the temperature of, a biological site during ablation.
- In one aspect, the invention relates to an apparatus for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature. The apparatus includes a plurality of electrodes positionable proximal the biological tissue and at least one temperature sensing device located on at least one of the electrodes. The temperature sensing device provides a temperature signal indicative of the temperature at the interface between the at least one electrode and the tissue. The apparatus also includes a generator that operates under the control of a processor to apply power to each of the electrodes. The power has an associated phase angle and, within a time duration, a plurality of alternating on periods and off periods, one set of adjacent on and off periods defining a duty cycle. The processor is adapted to, during the on period of each duty cycle, set the phase angles of the power provided to each electrode such that at least two of the electrodes are provided power with different phase angles. The processor is further adapted to, in response to the temperature signal, determine the temperature at the at least one electrode/tissue interface; compare the interface temperature to the target temperature and adjust the duty cycle of the power provided to the at least one electrode in response to the comparison.
- In a detailed aspect of the invention, the processor is adapted to increase or decrease the duty cycle of the power to the at least one electrode when the at least one electrode/tissue interface temperature is less than or greater than the target temperature. In another detailed facet the processor is adapted to set the power to the at least one electrode to zero when the at least one electrode/tissue interface temperature is a specified first number of degrees greater than the target temperature to thereby allow the electrode to cool off. The processor is further adapted to incrementally increase the power to the electrode until the interface temperature is near the target temperature. In another detailed aspect, the apparatus further includes a power interruption device connected to the processor. The processor is adapted to control the power interruption device to interrupt power to the at least one electrode when the at least one electrode/tissue interface temperature is a specified second number of degrees greater than the target temperature.
- In another facet, the invention relates to an apparatus for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature. The apparatus includes a catheter having a plurality of electrodes at its distal end. The distal end may be positioned so that the electrodes are located proximal the biological tissue. The apparatus also includes a plurality of temperature sensing devices. At least one temperature sensing device is located on a select number of the electrodes. These devices provide a temperature signal indicative of the temperature at the interface between the electrode and the tissue. The apparatus also includes a generator operating under the control of a processor to apply power to each of the electrodes. The power has an associated phase angle and, within a time duration, a plurality of alternating on periods and off periods. One set of adjacent on and off periods define a duty cycle. The processor is adapted to, in response to each of the temperature signals, determine the temperature at the electrode/tissue interface and regulate the temperature of a plurality of temperature zones. Each zone includes at least one combination electrode/temperature sensing device. The temperature is regulated by controlling the power to at least one electrode in the zone. The processor is further adapted to regulate the power to a plurality of phase groups. Each group includes at least one electrode. The power is regulated by controlling the phase of the power to each electrode in the group such that during the on portion of a duty cycle the phase in one group is different then the phase in another group.
- In another aspect, the invention relates to a method for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature. The method comprises positioning a plurality of electrodes proximal the biological tissue. At least one of the electrodes carries a temperature sensing device for providing a temperature signal indicative of the temperature at the interface between the electrode and the tissue. The method also includes providing power to each of the electrodes. The power has, within a time duration, a plurality of alternating on periods and off periods, wherein one set of adjacent on and off periods defines a duty cycle. Also included in the method is, during the on period of each duty cycle, providing power with different phase angles to at least two of the electrodes; determining the temperature at the at least one electrode/tissue interface in response to the temperature signal; comparing the at least one electrode/tissue interface temperature to the target temperature; and adjusting the duty cycle of the power provided to the at least one electrode in response to the comparison.
- In another aspect, the invention relates to a method for delivering energy to biological tissue while maintaining the temperature of the biological tissue near a target temperature. The method comprises positioning a catheter having a plurality of electrodes at its distal end so that the electrodes are located proximal the biological tissue. A select number of the electrodes carry at least one temperature sensing device. The device provides a temperature signal indicative of the temperature at the interface between the electrode and the tissue. The method also includes providing power having an associated phase and, within a time duration, a plurality of alternating on periods and off periods, one set of adjacent on and off periods defining a duty cycle. The method further includes, in response to the temperature signals, determining the temperature at the electrode/tissue interface; individually regulating the temperature of a plurality of temperature zones, wherein each zone comprises at least one combination electrode/temperature sensing device. The temperature is regulated by controlling the power to at least one electrode in the zone. The method further includes regulating the power to a plurality of phase groups, wherein each group comprises at least one electrode. The power is regulated by controlling the phase of the power to each electrode in the group such that during the on portion of a duty cycle the phase in one group is different then the phase in another group.
- These and other aspects and advantages of the present invention will become apparent from the following more detailed description, when taken in conjunction with the accompanying drawings which illustrate, by way of example, the preferred embodiments of the invention.
- FIG. 1 is a schematic diagram of an ablation apparatus including a power control system, electrode device and backplate;
- FIGS. 2-1 and 2-2 form a block diagram presenting more detail of a power control system in accordance with aspects of the invention, showing phase angle control, duty cycle control, and impedance and temperature monitoring;
- FIG. 3 is a diagram of a multi-channel ablation apparatus in accordance with aspects of the invention wherein a single microprocessor controls the phase angle and duty cycle of each channel individually;
- FIG. 4 depicts a first power waveform having a first phase angle and alternating instances of peak power and very low power;
- FIG. 5 depicts a second power waveform having a second phase angle different from the first phase angle and alternating instances of peak power and very low power;
- FIG. 6 presents a time frame (TF) diagram showing a fifty-percent duty cycle;
- FIG. 7A depicts the phase relationship and voltage potential between the first and second power waveforms having first and second phase angles respectively, as a function of time;
- FIG. 7B depicts the phase relationship and voltage potential between the first and second power waveforms having second and first phase angles respectively, as a function of time;
- FIGS. 8A, 8B, 8C, 8D, and 8E are schematic diagrams of an embodiment of a power control system in accordance with aspects of the invention with FIG. 8A showing how FIGS. 8B, 8C, 8D and 8E are related;
- FIG. 9A is a three dimensional representation of an ablation apparatus having a linear array of band electrodes in contact with a biological site with a backplate at the opposite side of the biological site, in which the phase angle difference between adjacent electrodes of the linear array is zero degrees;
- FIGS. 9B through 9D depict, along the x, y, and z axes shown, the depth of the lesions formed by the ablation apparatus of FIG. 9A showing that the apparatus acts as a unipolar device with multiple electrodes and the resulting lesions are discontinuous;
- FIG. 10A is a three dimensional representation of an ablation apparatus having a linear array of band electrodes in contact with a biological site with a backplate at the opposite side of the biological site, in which the phase angle difference between adjacent electrodes is 180 degrees;
- FIGS. 10B through 10D depict, along the x, y, and z axes shown, the continuity and depth of a lesion formed by the ablation apparatus of FIG. 10A showing that the apparatus acts as a bipolar device with no significant amount of current flowing to the backplate;
- FIG. 11A is a three dimensional representation of an ablation apparatus having a linear array of band electrodes in contact with a biological site with a backplate at the opposite side of the biological site, in which the phase difference between adjacent electrodes is approximately 90 degrees; and
- FIGS. 11B through 11D depict, along the x, y, and z axes shown, the continuity and depth of a lesion formed by the ablation apparatus of FIG. 11A showing the greater depth of lesion resulting from the phase angle difference.
- FIG. 12 presents a block diagram of the current flow among electrodes and the backplate through the biological site for adjacent electrodes having different phase angles;
- FIG. 13 presents the same block diagram as FIG. 12 with the phase angles between adjacent electrodes reversed; and
- FIGS. 14A through 14D present, along the x, y, and z axes shown, the increased continuity, depth, and uniformity of a lesion formed by the alternating phase apparatus and method shown in previous figures.
- Turning now to the drawings, in which like reference numerals are used to designate like or corresponding elements among the several figures, in FIG. 1 there is shown an
ablation apparatus 10 in accordance with aspects of the present invention. Theapparatus 10 includes apower control system 12 that provides power or drive 14 to anelectrode device 16. Thepower control system 12 comprises apower generator 18 that may have any number of output channels through which it provides thepower 14. The operation of thepower generator 18 is controlled by acontroller 20 which outputs control signals 21 to thepower generator 18. Thecontroller 20 monitors thepower 14 provided by thepower generator 18. In addition, thecontroller 20 also receives temperature signals 22 from theelectrode device 16. Based on thepower 14 andtemperature signals 22 thecontroller 20 adjusts the operation of thepower generator 18. Abackplate 24 is located proximal to thebiological site 26 opposite the site from theelectrode device 16, and is connected by abackplate wire 28 to thepower generator 18. Thebackplate 24 is set at the reference level to the power provided to the electrodes, as discussed in detail below. - The
electrode device 16 is typically part of asteerable EP catheter 30 capable of being percutaneously introduced into abiological site 26, e.g., the atrium or ventricle of the heart. Theelectrode device 16 is shown in schematic form with the components drawn to more clearly illustrate the relationship between the components and the relationship between the components and thepower control system 12. In this embodiment, thecatheter 30 comprises adistal segment 34 and ahandle 31 located outside the patient. A preferred embodiment of theelectrode device 16 includes twelveband electrodes 32 arranged in a substantially linear array along thedistal segment 34 of thecatheter 30. Theelectrode device 16 may include atip electrode 36. (For clarity of illustration, only fourband electrodes 32 are shown in the figures although as stated, a preferred embodiment may include many more.) Theband electrodes 32 are arranged so that there isspace 38 between adjacent electrodes. In one configuration of theelectrode device 16, the width of theband electrodes 32 is 3 mm and thespace 38 between the electrodes is 4 mm. The total length of theelectrode device 16, as such, is approximately 8 cm. - The arrangement of the
band electrodes 32 is not limited to a linear array and may take the form of other patterns. A substantially linear array is preferred for certain therapeutic procedures, such as treatment of atrial fibrillation, in which linear lesions of typically 4 to 8 cm in length are desired. A linear array is more easily carried by thecatheter 30 and also lessens the size of the catheter. - The
band electrodes 32 are formed of a material having a significantly higher thermal conductivity than that of thebiological tissue 26. Possible materials include silver, copper, gold, chromium, aluminum, molybdenum, tungsten, nickel, platinum, and platinum/10% iridium. Because of the difference in thermal conductivity between theelectrodes 32 and thetissue 26, theelectrodes 32 cool off more rapidly in the flowing fluids at the biological site. The power supplied to theelectrodes 32 may be adjusted during ablation to allow for the cooling of the electrodes while at the same time allowing for the temperature of the tissue to build up so that ablation results. Theelectrodes 32 are sized so that the surface area available for contact with fluid in the heart, e.g., blood, is sufficient to allow for efficient heat dissipation from the electrodes to the surrounding blood. In a preferred embodiment, theelectrodes 32 are 7 French (2.3 mm in diameter) with a length of 3 mm. - The thickness of the
band electrodes 32 also affects the ability of the electrode to draw thermal energy away from the tissue it contacts. In the present embodiment, theelectrodes 32 are kept substantially thin so that the electrodes effectively draw energy away from the tissue without having to unduly increase the outer diameter of the electrode. In a preferred embodiment of the invention, the thickness of the band electrodes is 0.05 to 0.13 mm (0.002 to 0.005 inches). - Associated with the
electrode device 16 aretemperature sensors 40 for monitoring the temperature of theelectrode device 16 at various points along its length. In one embodiment, eachband electrode 32 has atemperature sensor 40 mounted to it. Eachtemperature sensor 40 provides atemperature signal 22 to thecontroller 20 which is indicative of the temperature of therespective band electrode 32 at that sensor. In another embodiment of the electrode device 16 atemperature sensor 40 is mounted on everyother band electrode 32. Thus for a catheter having twelve electrodes, there are temperature sensors on six electrodes. In yet another embodiment of theelectrode device 16 every other electrode has twotemperature sensors 40. In FIG. 1, which shows an embodiment having one temperature sensor for each electrode, there is shown asingle power lead 15 for eachelectrode 32 to provide power to each electrode for ablation purposes and two temperature leads 23 for eachtemperature sensor 40 to establish the thermocouple effect. - In another approach, the drive wire may comprise one of the thermocouple wires or may comprise a common wire for a plurality of thermocouples mounted on the same electrode. The inventors hereby incorporates by reference U.S. Pat. Nos. 6,049,737 and 6,045,550 both of which are assigned to the assignee of the present invention.
- Turning now to FIGS. 2-1 and 2-2, a block diagram of an
ablation apparatus 10 and method in accordance with aspects of the invention is presented. In FIGS. 2-1 and 2-2, a single channel of thepower control system 12 is depicted. This channel controls the application of power to asingle electrode 32. As will be discussed in relation to other figures, a channel may control a plurality or group of electrodes. In FIG. 2-1, amicroprocessor 42, which is part of the controller 20 (FIG. 1), provides a dutycycle control signal 44 to a duty cycle generator (“DCG”) 45. In this case, theduty cycle generator 45 receives thecontrol signal 44 by an 8-bit latch 46. Thelatch 46 provides an 8-bit signal 47 to aduty cycle comparator 48. Thecomparator 48 compares the 8-bit signal 47 to a count from an 8-bitduty cycle counter 50 and if the count is the same, provides a duty cycle offsignal 49 to theduty cycle gate 52. Thegate 52 is connected to a frequency source (“FS”) 54, such as an oscillator that produces 500 kHz. When thegate 52 receives the duty cycle offsignal 49 from thecomparator 48, it stops its output of the frequency source signal through the gate and no output exists. - At a frequency of 500 kHz, an 8-bit control has a period or time frame of 0.5 msec. At a fifty-percent duty cycle, the electrode is in the off period only 0.25 msec. To allow for greater cooling of the electrode, the period or time frame 78 (FIG. 6) is lengthened by use of a
prescalar 56 interposed between thefrequency source 54 and thecounter 50. In one embodiment, theprescalar 56 lengthens the period to 4 msec thus allowing for a 2 msec off period during a fifty-percent duty cycle. This results in a sufficient cooling time for the very thin band electrodes discussed above. Other lengths of the period may be used depending on the circumstances. It has been found that a ten percent duty cycle is particularly effective in ablating heart tissue. The combination of the application of high peak power, a ten percent duty cycle, the use of high thermal conductivity material in the band electrodes, and fluids flowing past the band electrodes which have a cooling effect on the electrodes result in a much more effective application of power to the tissue. Ablation occurs much more rapidly. - A
terminal count detector 58 detects the last count of the period and sends aterminal count signal 59 to thegate 52 which resets the gate for continued output of the frequency source signal. This then begins the on period of the duty cycle and thecounter 50 begins its count again. In one preferred embodiment, the duty cycle is set at fifty percent and the 8-bit latch is accordingly set to 128. In another embodiment, the duty cycle is set at ten percent. - A programmable logic array (“PLA”) 60 receives phase control signals 61 from the
microprocessor 42 and controls the phase of thefrequency source 54 accordingly. In one embodiment, thePLA 60 receives theterminal count signal 59 from theterminal count detector 58 and only permits phase changes after receiving that terminal count signal. - The output signal from the
gate 52 during the on period of the duty cycle is provided to a binary power amplifier (“BPA”) 62 that increases the signal to a higher level, in this case, 24 volts. The amplified signals are then filtered with a band pass filter (“BPF”) 64 to convert the somewhat square wave to a sine wave. Theband pass filter 64 in one embodiment is centered at 500 kHz. The filtered signal is then provided to an isolated output transformer (“IOT”) 66 that amplifies the signal to a much higher level, for example 350 volts peak-to-peak. This signal is then sent to a relay interconnect (“RI”) 67 before it is provided as a poweroutput signal OUTn 14 to anelectrode 32 at the biological site to cause ablation. - The
power output signal 14 from theisolated output transformer 66 is monitored in one embodiment to determine the impedance at theelectrode 32. In the embodiment shown in FIGS. 2-1 and 2-2, a voltage and current monitor (“VCM”)68 is used. Themonitor signal 69 is converted to digital form by an A-to-D converter (“ADC”) 70 and provided to themicroprocessor 42. As previously mentioned, some or all of theelectrodes 32 may include a temperature sensor 40 (FIG. 1) that provides temperature signals 22 (FIG. 2-2) which are used to determine the temperature at theelectrode 32. In one embodiment of the invention, thepower 14, in conjunction with the temperature signals 22, are used to determine the temperature at theelectrode 32. Both the temperature signals 22 and thepower 14 pass through a temperature filter (“FL”) 73 before being sent to themicroprocessor 42. In the alternative, thetemperature filter 73 is contained in a printed circuit board separate from thecontroller 20 and contains its own processor. In either case, thefilter 73 filters out any RF noise present in thepower 14 so that the signal may be used for temperature monitoring purposes. In another embodiment, the microprocessor monitors thepower 14 andtemperature signals 22 only during the off periods of thepower 14 duty cycle. Accordingly, negligible RF noise is present in the power line and filtration is not necessary. In either embodiment, themicroprocessor 42 may alter the duty cycle of thepower 14 in response to either or both of the impedance or temperature signals. - In a manual arrangement, the temperature sensed and/or the determined impedance may be displayed to an operator. The operator in response may then manually control the duty cycle or other power parameters such as by rotating a knob mounted on a front panel of an instrument. In the case of a multiple channel instrument and catheter, as discussed below, multiple knobs may be provided in this manual arrangement for control over each channel.
- Referring now to FIG. 3, a multiple channel ablation apparatus is shown. Although only three complete channels are shown, the apparatus comprises many more as indicated by the successive dots. Those channels are not shown in FIG. 3 to preserve clarity of illustration. By providing different voltage levels between two
electrodes 32 in an array, current flows between those electrodes in a bipolar electrode approach. By setting the backplate 24 (FIG. 1) at a voltage level different from at least one of thoseelectrodes 32, current flows between that electrode and the backplate. By controlling the voltage levels among the three (two electrodes and backplate), the current flow through thebiological site 26 can be more precisely controlled. One technique for setting different voltage levels between theelectrodes 32 is to maintain a phase difference between them in an AC approach. By setting thebackplate 24 at the reference level, current flows between theelectrodes 32 and the backplate. - The
single microprocessor 42, which again is part of the controller 20 (FIG. 1), controls the duty cycle and the phase of each channel individually in this embodiment. Each channel shown comprises the same elements and each channel produces its own power output signal 14 (OUT1, OUT2, through OUTn where “n” is the total number of channels) on respective electrode leads (LEAD 1,LEAD 2, through LEAD n where “n” is the total number of leads) to theelectrodes 32. This multi-channel approach permits more individual control over each electrode. For example, the duty cycle of the power applied to each electrode can be individually controlled. One electrode may have a ten percent duty cycle while another has a thirty percent duty cycle. - Referring now to the first and second output signals OUT 1 and OUT2 of FIG. 3, the signals, as shown in FIGS. 4, 5, and 6, have alternating instances of peak power i.e., “on”
periods 74, and verylow power 76, i.e., “off” periods. Typically, theoutput power 14 is a 500 kHz sine wave. In FIGS. 4 and 5, the number of cycles of the sine wave contained within one onperiod 74 has been substantially reduced in the drawing to emphasize the phase difference between the first and second output signals OUT1, OUT2. Preferably, the voltage of eachpower signal 14 during anoff period 76 is substantially zero and during an onperiod 74 is approximately 350 volts peak-to-peak. - The power OUT 1 and OUT2 also have a variable duty cycle for controlling the length of the on
period 74 and the off-period 76 within a time frame 78 (see FIG. 6). The duty cycle is the ratio of the length of the onperiod 74 to the length of theentire time frame 78. The effective power is the peak power times the duty cycle. Thus, a signal having a peak power of 100 watts and a 50% duty cycle has an effective power of 50 watts. - As shown in FIGS. 4, 5, and 6, the two power signals OUT1, OUT2 are phased differently from each other. As discussed above, the phase angle of each power signal is set and controlled by the
processor 42 andPLA 60. Each power signal OUT1 and OUT2 has a respective phase angle and those phase angles differ between the two of them. The phase angle difference between the power OUT1 and OUT2 produces a voltage potential between the band electrodes 32 (FIG. 1) that receive the power. This voltage potential, in turn, induces current flow between theband electrodes 32. The phase angle relationship of the power and the voltage potential produced as a function of time is shown in FIGS. 7A and 7B. The potential between electrodes Ve-e is defined by: - where: ΔΦ=phase angle difference between electrodes
- V=voltage amplitude of power
- f=frequency in hertz
- t=time
- FIG. 7A shows first and second power OUT 1 and OUT2 provided to first and second electrodes respectively having a phase angle difference ΔΦ with OUT1 leading OUT2 by 132 degrees. FIG. 7B shows the same power OUT1 and OUT2 but with the phase angles reversed where OUT2 is now leading OUT 1 by 132 degrees.
- With reference now to FIGS. 8A through 8E, schematic diagrams of an embodiment of the
ablation apparatus 10 of FIGS. 2-1 and 2-2 are presented in FIGS. 8B through 8E while FIG. 8A shows how FIGS. 8B through 8E should be oriented in relation to each other. Thefrequency source 54 provides asignal 80, typically at 500 kHz with a phase angle controlled by themicroprocessor 42 through thePLA 60, to theduty cycle generator 45. Theduty cycle generator 45 modulates thefrequency source signal 80 to produce the selected duty cycle in accordance with the dutycycle control signal 44 as previously described. Theduty cycle generator 45 outputs twosignals binary power amplifier 62. A dual MOSFET driver U2 receives the signals, converts their 5V level to a 12V level, and sends each to a transformer T2 which transforms the signals into 24 V peak-to-peak power. - The 24V power is then sent to a
multi-state driver 86 which includes a configuration of FETs Q2, Q3, Q4, and Q5. During a conducting state of thedriver 86, which is typically the onperiod 74 of the power, these FETs Q2 through Q5 conduct and forward the power to abandpass filter 64 comprising a series LC network. During a high-impedance state of thedriver 86, which is typically during theoff period 76 of the power, the FETs Q2 through Q5 are nonconducting and no power is sent to thebandpass filter 64. Instead the FETs Q2 through Q5 present a high impedance load to any signals received through theelectrode 32. Typically the load impedance on the FETs Q2 through Q5 presented by the circuit following the FETs, the electrode, and the tissue is approximately 150 Ω but transformed through the output transformer T3, it presents a load impedance to the FETs Q2-Q5 of approximately 0.5 to 1 Ω. In the off state, the FETs present an impedance of approximately 250 Ω which is large in comparison to the transformed load impedance of approximately 0.5 to 1 Ω. Therefore, very little power flows when the FETs are in the off state. - The
bandpass filter 64 operates to shape the output signal provided by thebinary amplifier 62 from a square wave to a sinusoidal wave. The filteredsignal 85 then passes to theisolated output section 66 where it is step-up transformed to 350 volt peak-to-peak sinusoidal power at T3. The power is then split into two identical power signals OUT1A, OUT1B and provided to two or morerespective band electrodes 32 on the output lines LEAD1A, LEAD1B. - The
isolated output section 66 also includesrelays 88 that may be individually opened to remove the power signals OUT1A, OUT1B from the electrode leadsLEAD 1A,LEAD 1B when an alert condition is detected, such as high temperature or high impedance at therespective electrode 32. As previously mentioned these conditions are determined by themicroprocessor 42 which receives signals indicative of the temperature and impedance at each of theband electrodes 32. - The power from the
isolated output section 66 is monitored and representative signals are supplied to an RF voltage andcurrent monitor 68 where in this case, the voltage and current of each output signal are measured to determine the impedance of the particular channel. The measured signals are sent to an A-to-D converter 70 (FIG. 2-2) before being sent to themicroprocessor 42 for impedance monitoring. If the impedance is above a threshold level indicative of blood clotting or boiling, themicroprocessor 42 sends a signal to theduty cycle generator 45 to reduce or discontinue the duty cycle of the power OUT1A, OUT1B and thus lower the effective power delivered to theband electrodes 32. - Similarly, the temperature at the
electrodes 32 is determined by monitoring thepower 14 andtemperature signals 22 and measuring the voltage difference between the signals. As previously mentioned, in one embodiment of the invention, these signals pass through a filter 73 (FIG. 2-2) before being sent to themicroprocessor 42. The voltage value is converted to a temperature and if the temperature is above a threshold level the duty cycle of thepower 14 is reduced. In the case where a single lead is used to provide a signal which is used to determine the temperature as well as provide power to theelectrode 32, the signal from the lead is received on temperature leads 87, 89 connected at the output side of therelays 88. - As shown in FIG. 3, the duty cycle of each
electrode 32 may be individually controlled by themicroprocessor 42. As previously mentioned, based on the temperature at anelectrode 32 and the current and voltage of the output signal provided to an electrode, the duty cycle of the output signal may be adjusted. For example, oneelectrode 32 may have a temperature requiring a duty cycle of ten percent, while another electrode may have a temperature which allows for a fifty percent duty cycle. In an embodiment in which everyother electrode 32 has atemperature sensor 40, the electrodes are grouped in pairs with each electrode in the pair having the same duty cycle. - In operation, as depicted in FIGS. 9A through 11D, the
electrode device 16 and thebackplate 24 are positioned proximal thebiological site 26 undergoing ablation such that the biological site is interposed between the electrode device and the backplate. The band electrodes 32 (only one of which is indicated by a numeral 32 for clarity of illustration) of theelectrode device 16 each receives power OUT1, OUT2, OUT3, OUT4 having a phase angle onLEAD 1 throughLEAD 4. In one embodiment, everyother electrode 32 receives the same phase angle. Therefore, the phase angle of electrode A equals the phase angle of electrode C and the phase angle of electrode B equals the phase angle of electrode D. The advantages of this arrangement are described below. In a preferred embodiment, theelectrodes 32 are formed into a linear array as shown. In addition, athermocouple temperature sensor 40 is located at each of the electrodes A, B, C, and D and uses the electrode power lead LEADS 1 through 4 as one of the sensor leads. Thesensors 40 provide temperature sensor signals 22 for receipt by thepower control system 12. - In another embodiment,
alternate electrodes 32 may be grouped together and each may receive the same power having the same phase angle and duty cycle. Another group or groups ofelectrodes 32 may be interspaced with the first group such that the electrodes of one group alternate with the electrodes of the other group or groups. Eachelectrode 32 in a particular group of electrodes has the same phase angle and duty cycle. For example, electrodes A and C may be connected to the same power while interspaced electrodes B and D may be connected to a different power output signal. - The use of individual power signals also provides the ability to disable any combination of
electrodes 32 and thereby effectively change the length of theelectrode device 16. For example, in one configuration of the present invention anelectrode device 16 with twelveelectrodes 32 receives twelve power signals from a twelve channelpower control system 12. Theelectrodes 32 are 3 mm in length and are 4 mm apart. Accordingly, by disabling various electrodes, a virtual electrode of any length from 3 mm to 8 cm may be produced by theelectrode device 16. In either arrangement thebackplate 24 is maintained at the reference voltage level in regard to the voltage level of the power OUT1 through OUTn. - As previously described, by varying the phase angles between the power OUT 1, OUT2 supplied to each
electrode 32, a phase angle difference is established between adjacent band electrodes. This phase angle difference may be adjusted to control the voltage potential betweenadjacent band electrodes 32 and thus to control the flow of current through thebiological site 26. The flow of current Ie-e betweenadjacent band electrodes 32 is defined by: - where: ΔΩ=phase angle difference between electrodes
- V=voltage amplitude of power
- Z e-e=impedance between electrodes
- f=frequency in hertz
- t=time
-
- where: ΔΩ=phase angle difference between electrodes
- V=voltage amplitude of power
- Z e-b=impedance between electrode and backplate
- f=frequency in hertz
- t=time
-
- where: ΔΩ=phase angle difference between electrodes
- FIGS. 9A through 11D illustrate various current flow patterns within a biological site. The depths and widths of the lesions depicted in FIGS. 9 through 11 are not necessarily to scale or in scalar proportion to each other but are provided for clarity in discerning the differences between the various power application techniques. When the phase difference between
adjacent electrodes 32 is zero degrees, no current flows between the electrodes in accordance with Eq. 2 above, and the apparatus operates in a unipolar fashion with the current flowing to thebackplate 24 as shown in FIGS. 9A through 9D. Substantially all current flows from theband electrodes 32 to thebackplate 24 forming a series of relatively deep,acute lesions 90 along the length of theelectrode device 16. As seen in the top view of FIG. 9B and the side view of FIG. 9D, the lesions are discrete. Thelesions 90 are discontinuous in regard to each other. - When the phase difference between
adjacent electrodes 32 is 180 degrees the apparatus operates in both a unipolar and bipolar fashion and the current flow pattern is as shown in FIG. 10A. With this phase difference, approximately twice as much current flows betweenadjacent band electrodes 32 than flows from the band electrodes to thebackplate 24. The resultinglesion 92 is shallow but is continuous along the length of theelectrode device 16. The continuity and shallow depth of thelesion 92 are illustrated in FIGS. 10B through 10D. Nevertheless, the lesion depth is still greater than that created by prior bipolar ablation methods alone. - When the phase difference between
adjacent electrodes 32 is set within the range of a value greater than zero to less than 180 degrees, the current flow varies from a deep, discontinuous unipolar pattern to a more continuous, shallow bipolar pattern. For example, when the phase difference betweenadjacent electrodes 32 is around 90 degrees, the current flows as shown in FIG. 11A. With this phase difference, current flows betweenadjacent band electrodes 32 as well as between the band electrodes and thebackplate 24. Accordingly, a lesion which is both deep and continuous along the length of theelectrode device 16 is produced. The continuity and depth of thelesion 94 is illustrated in FIGS. 11B through 11D. In one embodiment of FIG. 1A, adjacent electrodes alternated in phase but were provided with power in groups. Electrodes A and C were provided with power at a first phase angle and electrodes B and D were provided with power at a second phase angle, different from the first. - Thus, in accordance with the present invention the phase angle of the power may be adjusted in order to produce a lesion having different depth and continuity characteristics. In selecting the phase angle difference necessary to produce a continuous lesion having the greatest possible depth, other elements of the
electrode device 16 are considered. For example, the width of theband electrodes 32 and the spacing between the electrodes are factors in selecting an optimum phase angle. In a preferred embodiment of the present invention, as pointed out above, the width of the band electrodes is 3 mm, the spacing between the electrodes is 4 mm and the electrodes receive power which establish a phase difference of 132 degrees between adjacent electrodes. With this configuration a long continuous lesion having a length of between approximately 3 mm and 8 cm and a depth of 5 mm or greater was produced depending on the number of electrodes energized, the duty cycle employed, and the duration of power application. - In another embodiment of the invention, energy is applied to the
biological tissue 26 during the on period of the duty cycle in an alternating unipolar-bipolar manner. During the unipolar mode segment a voltage potential is established between theelectrodes 32 and thebackplate 24. Thus current flows through thetissue 26 between theelectrodes 32 and thebackplate 24. - During the bipolar mode segment a voltage potential is established between at least two of the
electrodes 32 rather than between the electrodes and thebackplate 24. Thus current flows through thetissue 26 between theelectrodes 32. While operating in this mode the voltage difference between theelectrodes 32 may be established by providing power with different phase angles to the electrodes as previously mentioned. Alternatively, some of theelectrodes 32 may be connected to a reference potential while others are maintained at a different voltage level. - By adjusting the duration of the unipolar and bipolar mode segments within the on period of the duty cycle, the continuity and depth of the lesion produced may be controlled. For example, operating in the unipolar mode for one-fourth of the on period and in the bipolar mode for three-fourths of the on period produces a lesion having a continuity and depth similar to the
lesion 94 illustrated in FIGS. 11B through 11D. - Referring to FIGS. 8B through and 8E, the following devices are shown:
Device Part No. Manufacturer U1 GAL6002B Lattice U2 SN75372 numerous Q1 1RFZ34N numerous Q2, Q3, Q4, Q5 1RFZ44N numerous Q7, Q8, Q9 MPF6601 numerous R3, R5 1 Ω numerous T1, T4 CMI-4810 Corona Magnetics, Inc. T2 GFS97-0131-1 GFS Manufacturing T5 CMI-4809 Corona Magnetics, Inc. - The transformer denoted by “T 3” is a 1:12 turns ratio, single turn primary, step up transformer wound on a TDK core PC50EER23Z.
- FIG. 12 presents a block diagram of the current flow among
electrodes 32 and thebackplate 24 through thebiological site 26 for adjacent electrodes having different phase angles where the phase angles of the A and C electrodes lead the phase angles of the B and D electrodes. It has been noted that with the approach shown in FIG. 12, the vector sum of the currents flowing through thesite 26 is such that more current flows at one or more electrodes than at others. This is shown figuratively with shorter arrows leading to the backplate from the B and D electrodes. Although the ablation volume is greater than in the prior techniques, the ablation volume appears irregular or nonuniform as shown in FIG. 11D. It is desirable to have a more uniform ablation volume, especially as to depth, so that irregular electrical signals do not pass under the ablation volume at a point having less depth and require a repeat of the ablation procedure. - FIG. 13 presents the same block diagram as FIG. 12 with the phase angles between adjacent electrodes reversed. In FIG. 13, the phase angles of the power at the B and
D electrodes 32 now lead the phase angles of the power at the A andC electrodes 32. The change in current flow due to this opposite phasing is represented figuratively with shorter arrows now at the A and C electrodes thus balancing the current flow pattern of FIG. 12. It has been found that by alternating the phase angles such as shown in FIGS. 12 and 13, a much more uniform current flow and much more uniform ablation volume result. A cumulative effect of the current flow causes the tissue between all theband electrodes 32 and thebackplate 24 to become ablated, depth-wise through thebiological site 26, at a substantially even rate and thus a lesion having substantially uniform depth is produced. This is shown in FIGS. 14A through 14D where anablation volume 96 is shown, which has much greater uniformity in shape. In particular, theablation lesion 96 has a uniform depth and gives rise to a high level of confidence that the ablation volume created with the ablation apparatus in accordance with the invention will successfully destroy the tissue causing the arrhythmia. - In one embodiment, the phase between the electrodes was alternated as shown in FIGS. 12 and 13 only during the off period of the duty cycle. That is, and with reference to FIG. 6, during the entire on
period 74 of the duty cycle of onetime frame 78, the phase angles of the power at the A andC electrodes 32 led the phase angles of the power at the B andD electrodes 32 by 132 degrees. During the following offperiod 76 of thesame time frame 78, the phase angles of the power to be supplied was changed to be opposite those phase angles used during the onperiod 74, in preparation for the next on period. Then at the next onperiod 74, the phase angles of the power provided to electrodes B and D led the phase angles of the power provided to the A and C electrodes by 132 degrees during that entire on period. During the immediately subsequent off period, the phase angles were again changed so that electrodes A and C would lead electrodes B and D. - During an ablation procedure, the
processor 42 monitors the temperatures at the electrodes/tissue interface and automatically makes power adjustments in view of these temperatures. As previously mentioned, in order to effectively ablate tissue it is desirable to uniformly elevate the tissue temperature to a target ablation temperature. For biological tissue this target temperature may range from 50° C. to 65° C. - In one configuration of the system, the temperature of each electrode/tissue interface is monitored and the power applied to each
electrode 32 is individually adjusted to maintain the electrode/tissue interface temperature at or near the target temperature. An interface temperature within 5° C. degrees of the target temperature is considered to be “near” the target temperature. Adjustments to the power are made by changing the duty cycle of the power being applied to theelectrode 32. When the electrode/tissue interface temperature is less than the target temperature, the duty cycle is increased. Conversely, when the interface temperature is greater than the target temperature, the duty cycle is decreased. - Under certain electrode/tissue interface temperature conditions, the
processor 42 may institute a “soft cutoff.” During a soft cutoff the processor temporarily sets the power to anelectrode 32 to zero by setting the duty cycle of the power such that there is no on portion of the duty cycle. During this continuous off period, the electrode/tissue interface is able to cool off to a temperature less than the temperature that caused the cutoff and usually to a temperature less than the target temperature. Once the interface cools off, theprocessor 42 incrementally increases the power to theelectrode 32 until the interface temperature settles to a temperature near the target temperature. The incremental increase in power may be achieved by programming the processor, using well known proportional integral derivative (PID) algorithms, to increase the duty cycle in either a step like or smooth, continuous manner. Conditions which necessitate a soft cutoff may be programmed into theprocessor 42. Such condition may include an interface temperature that is greater than the target temperature by a first specified number of degrees. For example, if the target temperature is 65° C. and the measured interface temperature is at least 5 degrees greater than 65° C., then the electrode may be subject to a soft cutoff. - Under more severe electrode/tissue interface temperature conditions, the
processor 42 may institute a “hard cutoff.” During a hard cutoff, power to anelectrode 32 may be interrupted by opening the relay 88 (FIG. 80) associated with the power output providing the power to theelectrode 32. Conditions which necessitate a hard cutoff may be programmed into theprocessor 42. Such conditions may include an interface temperature that is greater than the target temperature by a second specified number of degrees that is greater than the specified number of degrees associated with a soft cutoff. For example, if the target temperature is 65° C. and the measured interface temperature is at least 10 degrees greater than 65° C., then the power to the electrode may be interrupted. - In another configuration of the system, the
electrodes 32 are grouped into electrode zones, with each zone including one or more electrodes. In a preferred embodiment, the electrodes within a zone are adjacent each other. At least one of theelectrodes 32 within each zone includes atemperature sensor 40. Theprocessor 42 monitors the interface temperatures of eachelectrode 32 having atemperature sensor 40 and adjusts the power to each of the electrodes within an electrode zone based on the maximum interface temperature in that zone. Accordingly, if theelectrode 32 having the maximum interface temperature provides an interface temperature sufficient to warrant a duty cycle increase, duty cycle decrease, soft cutoff or hard cutoff, eachelectrode 32 within the zone is subject to the same adjustment. - The inventors hereby incorporate by reference application Ser. No. 09/073,112, now U.S. Pat. No. 6,171,305 and U.S. Pat. No. 6,050,994 both of which are assigned to the assignee of the present invention.
- While several particular forms of the invention have been illustrated and described, it will be apparent that various modifications can be made without departing from the spirit and scope of the invention. For example, the
controller 20 is shown in FIG. 1 as forming a part of thepower control system 12. However, it may take other forms such as an external processor in a separate computer for example. Likewise, duty cycle control and phase control may be performed by circuits other than those shown here. Accordingly, it is not intended that the invention be limited, except as by the appended claims.
Claims (20)
1. A system for delivering energy to biological tissue, said system comprising:
a plurality of electrodes adapted to be positioned at the biological tissue;
at least one temperature sensing device located on at least one of the electrodes for providing a temperature signal indicative of the temperature at the interface between the at least one electrode and the tissue; and
a power control system adapted to:
apply power to each of the electrodes such that a voltage potential is established between at least two of the electrodes, the power having a duty cycle;
in response to the temperature signal, determine the temperature at the at least one electrode/tissue interface;
compare the interface temperature to a target temperature; and
adjust the duty cycle of the power provided to the at least one electrode in response to the comparison.
2. The system of claim 1 wherein the power control system is adapted to increase the duty cycle of the power to the at least one electrode when the at least one electrode/tissue interface temperature is less than the target temperature.
3. The system of claim 1 wherein the power control system is adapted to decreases the duty cycle of the power to the at least one electrode when the at least one electrode/tissue interface temperature is greater than the target temperature.
4. The system of claim 1 wherein the power control system is adapted to:
set the power to the at least one electrode to zero when the at least one electrode/tissue interface temperature is a specified first number of degrees greater than the target temperature; and
incrementally increase the power level to the at least one electrode until the interface temperature is near the target temperature.
5. The system of claim 4 further comprising:
a power interruption device connected to the power control system;
wherein the power control system is further adapted to control the power interruption device to interrupt power to the at least one electrode when the at least one electrode/tissue interface temperature is a specified second number of degrees greater than the target temperature, the second number of degrees being greater than the first number of degrees.
6. The system of claim 1 wherein:
the system comprises a plurality of temperature sensing devices, at least one device associated with each of the electrodes, each device for providing a temperature signal to the power control system indicative of the temperature at the associated electrode; and
the power control system provides separate power to each of the plurality of electrodes with the power being individually controllable as to duty cycle and the duty cycle of each separate power is controlled in response to the temperature signal from the associated electrode.
7. The system of claim 7 wherein the power has an associated phase angle and the voltage potential is established between the at least two electrodes by providing power with different phase angles to the electrodes.
8. A method of delivering energy to biological tissue, said method comprising:
positioning a plurality of electrodes adjacent the biological tissue, at least one of the electrodes carrying a temperature sensing device for providing a temperature signal indicative of the temperature at the interface between the electrode and the tissue;
establishing a voltage potential between at least two of the electrodes using duty cycled power;
determining the temperature at the at least one electrode/tissue interface in response to the temperature signal;
comparing the at least one electrode/tissue interface temperature to the target temperature; and
adjusting the duty cycle of the power provided to the at least one electrode in response to the comparison.
9. The method of claim 8 wherein positioning a plurality of electrodes comprises positioning a catheter carrying the electrodes proximal the tissue such that a select number of the plurality of electrodes are in contact with the tissue.
10. The method of claim 8 further comprising increasing the duty cycle of the power to the at least one electrode when the at least one electrode/tissue interface temperature is less than the target temperature.
11. The method of claim 8 further comprising decreasing the duty cycle of the power to the at least one electrode when the at least one electrode/tissue interface temperature is greater than the target temperature.
12. The method of claim 8 further comprising:
setting the power to the at least one electrode to zero when the at least one electrode/tissue interface temperature is a specified first number of degrees greater than the target temperature; and
incrementally increasing the power level to the at least one electrode until the interface temperature is near the target temperature.
13. The method of claim 12 further comprising interrupting the power to the at least one electrode when the at least one electrode/tissue interface temperature is a specified second number of degrees greater than the target temperature, the second number of degrees being greater than the first number of degrees.
14. The method of claim 8 wherein each of the electrodes carries a temperature sensing device and the method further comprises:
providing separate power to each of the plurality of electrodes; and
individually controlling the duty cycle of each separate power in response to the temperature signal from the electrode.
15. The method of claim 8 wherein the power has an associated phase angle and establishing the voltage potential between the at least two electrodes comprises providing power with different phase angles to the electrodes.
16. A system for delivering energy to biological tissue, said system comprising:
a plurality of electrodes adapted to be positioned at the biological tissue;
at least one temperature sensing device located on at least one of the electrodes for providing a temperature signal indicative of the temperature at the interface between the at least one electrode and the tissue; and
a power control system adapted to:
apply power to each of the electrodes such that at least two of the electrodes are provided power with different phase angles;
in response to the temperature signal, determine the temperature at the at least one electrode/tissue interface;
compare the interface temperature to a target temperature; and
adjust the power provided to the at least one electrode in response to the comparison.
17. The system of claim 16 wherein the power control system is adapted to increase the power to the at least one electrode when the at least one electrode/tissue interface temperature is less than the target temperature.
18. The system of claim 16 wherein the power control system is adapted to decreases the power to the at least one electrode when the at least one electrode/tissue interface temperature is greater than the target temperature.
19. The system of claim 16 wherein the power control system is adapted to:
set the power to the at least one electrode to zero when the at least one electrode/tissue interface temperature is a specified first number of degrees greater than the target temperature; and
incrementally increase the power to the at least one electrode until the interface temperature is near the target temperature.
20. The system of claim 19 further comprising:
a power interruption device connected to the power control system;
wherein the power control system is further adapted to control the power interruption device to interrupt power to the at least one electrode when the at least one electrode/tissue interface temperature is a specified second number of degrees greater than the target temperature, the second number of degrees being greater than the first number of degrees.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/400,770 US20030195501A1 (en) | 1998-05-05 | 2003-03-27 | RF ablation system and method having automatic temperature control |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US09/073,392 US6059778A (en) | 1998-05-05 | 1998-05-05 | RF ablation apparatus and method using unipolar and bipolar techniques |
| US09/501,472 US6200314B1 (en) | 1998-05-05 | 2000-02-09 | RF ablation apparatus and method using unipolar and bipolar techniques |
| US09/738,032 US6558378B2 (en) | 1998-05-05 | 2000-12-13 | RF ablation system and method having automatic temperature control |
| US10/400,770 US20030195501A1 (en) | 1998-05-05 | 2003-03-27 | RF ablation system and method having automatic temperature control |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/738,032 Continuation US6558378B2 (en) | 1998-05-05 | 2000-12-13 | RF ablation system and method having automatic temperature control |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20030195501A1 true US20030195501A1 (en) | 2003-10-16 |
Family
ID=24966286
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/738,032 Expired - Lifetime US6558378B2 (en) | 1998-05-05 | 2000-12-13 | RF ablation system and method having automatic temperature control |
| US10/400,770 Abandoned US20030195501A1 (en) | 1998-05-05 | 2003-03-27 | RF ablation system and method having automatic temperature control |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US09/738,032 Expired - Lifetime US6558378B2 (en) | 1998-05-05 | 2000-12-13 | RF ablation system and method having automatic temperature control |
Country Status (3)
| Country | Link |
|---|---|
| US (2) | US6558378B2 (en) |
| AU (1) | AU2002230812A1 (en) |
| WO (1) | WO2002047565A2 (en) |
Cited By (101)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060223455A1 (en) * | 2005-03-31 | 2006-10-05 | Silicon Laboratories Inc. | System and method for wireless communication using low-pulling digital interface signals |
| US20070167940A1 (en) * | 2003-03-28 | 2007-07-19 | Debbie Stevens-Wright | Method and apparatus for selecting operating parameter values in electrophysiology procedures |
| US20080300590A1 (en) * | 2006-12-07 | 2008-12-04 | Cierra, Inc. | Apparatus and methods for multipolar tissue welding |
| US20100030210A1 (en) * | 2008-08-01 | 2010-02-04 | Paulus Joseph A | Polyphase Electrosurgical System and Method |
| WO2010051305A1 (en) * | 2008-10-28 | 2010-05-06 | Smith & Nephew, Inc. | Electrosurgical device with controllable electric field profile |
| US7742795B2 (en) * | 2005-03-28 | 2010-06-22 | Minnow Medical, Inc. | Tuned RF energy for selective treatment of atheroma and other target tissues and/or structures |
| US7846158B2 (en) * | 2006-05-05 | 2010-12-07 | Covidien Ag | Apparatus and method for electrode thermosurgery |
| US7850685B2 (en) | 2005-06-20 | 2010-12-14 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US20110218526A1 (en) * | 2010-03-03 | 2011-09-08 | Medtronic Ablation Frontiers, Llc | Variable-output radiofrequency ablation power supply |
| US8273084B2 (en) | 2004-11-24 | 2012-09-25 | Medtronic Ablation Frontiers Llc | Atrial ablation catheter and method of use |
| US8295902B2 (en) | 2008-11-11 | 2012-10-23 | Shifamed Holdings, Llc | Low profile electrode assembly |
| US8361068B2 (en) | 2000-03-06 | 2013-01-29 | Medtronic Advanced Energy Llc | Fluid-assisted electrosurgical devices, electrosurgical unit with pump and methods of use thereof |
| US8396548B2 (en) | 2008-11-14 | 2013-03-12 | Vessix Vascular, Inc. | Selective drug delivery in a lumen |
| US8401667B2 (en) | 2008-11-17 | 2013-03-19 | Vessix Vascular, Inc. | Selective accumulation of energy with or without knowledge of tissue topography |
| US8486063B2 (en) | 2004-10-14 | 2013-07-16 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US8496653B2 (en) | 2007-04-23 | 2013-07-30 | Boston Scientific Scimed, Inc. | Thrombus removal |
| US8551096B2 (en) | 2009-05-13 | 2013-10-08 | Boston Scientific Scimed, Inc. | Directional delivery of energy and bioactives |
| US8617152B2 (en) | 2004-11-15 | 2013-12-31 | Medtronic Ablation Frontiers Llc | Ablation system with feedback |
| US8641704B2 (en) | 2007-05-11 | 2014-02-04 | Medtronic Ablation Frontiers Llc | Ablation therapy system and method for treating continuous atrial fibrillation |
| US8657814B2 (en) | 2005-08-22 | 2014-02-25 | Medtronic Ablation Frontiers Llc | User interface for tissue ablation system |
| US20140067029A1 (en) * | 2012-08-28 | 2014-03-06 | Boston Scientific Scimed, Inc. | Renal nerve modulation and ablation catheter electrode design |
| US8834461B2 (en) | 2005-07-11 | 2014-09-16 | Medtronic Ablation Frontiers Llc | Low power tissue ablation system |
| US20140296841A1 (en) * | 2010-01-25 | 2014-10-02 | Covidien Lp | System and method for monitoring ablation size |
| US8880185B2 (en) | 2010-06-11 | 2014-11-04 | Boston Scientific Scimed, Inc. | Renal denervation and stimulation employing wireless vascular energy transfer arrangement |
| US8920414B2 (en) | 2004-09-10 | 2014-12-30 | Vessix Vascular, Inc. | Tuned RF energy and electrical tissue characterization for selective treatment of target tissues |
| US8951251B2 (en) | 2011-11-08 | 2015-02-10 | Boston Scientific Scimed, Inc. | Ostial renal nerve ablation |
| US8974451B2 (en) | 2010-10-25 | 2015-03-10 | Boston Scientific Scimed, Inc. | Renal nerve ablation using conductive fluid jet and RF energy |
| US9005194B2 (en) | 2004-11-24 | 2015-04-14 | Medtronic Ablation Frontiers Llc | Atrial ablation catheter adapted for treatment of septal wall arrhythmogenic foci and method of use |
| US9023034B2 (en) | 2010-11-22 | 2015-05-05 | Boston Scientific Scimed, Inc. | Renal ablation electrode with force-activatable conduction apparatus |
| US9028485B2 (en) | 2010-11-15 | 2015-05-12 | Boston Scientific Scimed, Inc. | Self-expanding cooling electrode for renal nerve ablation |
| US9028472B2 (en) | 2011-12-23 | 2015-05-12 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9050106B2 (en) | 2011-12-29 | 2015-06-09 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US9060761B2 (en) | 2010-11-18 | 2015-06-23 | Boston Scientific Scime, Inc. | Catheter-focused magnetic field induced renal nerve ablation |
| US9079000B2 (en) | 2011-10-18 | 2015-07-14 | Boston Scientific Scimed, Inc. | Integrated crossing balloon catheter |
| US9084609B2 (en) | 2010-07-30 | 2015-07-21 | Boston Scientific Scime, Inc. | Spiral balloon catheter for renal nerve ablation |
| US9089350B2 (en) | 2010-11-16 | 2015-07-28 | Boston Scientific Scimed, Inc. | Renal denervation catheter with RF electrode and integral contrast dye injection arrangement |
| US9119600B2 (en) | 2011-11-15 | 2015-09-01 | Boston Scientific Scimed, Inc. | Device and methods for renal nerve modulation monitoring |
| US9119632B2 (en) | 2011-11-21 | 2015-09-01 | Boston Scientific Scimed, Inc. | Deflectable renal nerve ablation catheter |
| US9125667B2 (en) | 2004-09-10 | 2015-09-08 | Vessix Vascular, Inc. | System for inducing desirable temperature effects on body tissue |
| US9125666B2 (en) | 2003-09-12 | 2015-09-08 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation of atherosclerotic material |
| US20150265333A1 (en) * | 2012-10-25 | 2015-09-24 | Kyong-Min Shin | System for ablation utilizing multiple electrodes and method for controlling same |
| US9155589B2 (en) | 2010-07-30 | 2015-10-13 | Boston Scientific Scimed, Inc. | Sequential activation RF electrode set for renal nerve ablation |
| US9162046B2 (en) | 2011-10-18 | 2015-10-20 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US9173696B2 (en) | 2012-09-17 | 2015-11-03 | Boston Scientific Scimed, Inc. | Self-positioning electrode system and method for renal nerve modulation |
| US9186209B2 (en) | 2011-07-22 | 2015-11-17 | Boston Scientific Scimed, Inc. | Nerve modulation system having helical guide |
| US9186210B2 (en) | 2011-10-10 | 2015-11-17 | Boston Scientific Scimed, Inc. | Medical devices including ablation electrodes |
| US9192790B2 (en) | 2010-04-14 | 2015-11-24 | Boston Scientific Scimed, Inc. | Focused ultrasonic renal denervation |
| US9192435B2 (en) | 2010-11-22 | 2015-11-24 | Boston Scientific Scimed, Inc. | Renal denervation catheter with cooled RF electrode |
| US9220561B2 (en) | 2011-01-19 | 2015-12-29 | Boston Scientific Scimed, Inc. | Guide-compatible large-electrode catheter for renal nerve ablation with reduced arterial injury |
| US9220558B2 (en) | 2010-10-27 | 2015-12-29 | Boston Scientific Scimed, Inc. | RF renal denervation catheter with multiple independent electrodes |
| US9265969B2 (en) | 2011-12-21 | 2016-02-23 | Cardiac Pacemakers, Inc. | Methods for modulating cell function |
| US9277955B2 (en) | 2010-04-09 | 2016-03-08 | Vessix Vascular, Inc. | Power generating and control apparatus for the treatment of tissue |
| US9297845B2 (en) | 2013-03-15 | 2016-03-29 | Boston Scientific Scimed, Inc. | Medical devices and methods for treatment of hypertension that utilize impedance compensation |
| US9326751B2 (en) | 2010-11-17 | 2016-05-03 | Boston Scientific Scimed, Inc. | Catheter guidance of external energy for renal denervation |
| US9358365B2 (en) | 2010-07-30 | 2016-06-07 | Boston Scientific Scimed, Inc. | Precision electrode movement control for renal nerve ablation |
| US9364284B2 (en) | 2011-10-12 | 2016-06-14 | Boston Scientific Scimed, Inc. | Method of making an off-wall spacer cage |
| US9408661B2 (en) | 2010-07-30 | 2016-08-09 | Patrick A. Haverkost | RF electrodes on multiple flexible wires for renal nerve ablation |
| US9420955B2 (en) | 2011-10-11 | 2016-08-23 | Boston Scientific Scimed, Inc. | Intravascular temperature monitoring system and method |
| US9433760B2 (en) | 2011-12-28 | 2016-09-06 | Boston Scientific Scimed, Inc. | Device and methods for nerve modulation using a novel ablation catheter with polymeric ablative elements |
| US9463062B2 (en) | 2010-07-30 | 2016-10-11 | Boston Scientific Scimed, Inc. | Cooled conductive balloon RF catheter for renal nerve ablation |
| US9579030B2 (en) | 2011-07-20 | 2017-02-28 | Boston Scientific Scimed, Inc. | Percutaneous devices and methods to visualize, target and ablate nerves |
| WO2017049313A1 (en) * | 2015-09-17 | 2017-03-23 | Baylor College Of Medicine | Esophageal probes and methods |
| US9649156B2 (en) | 2010-12-15 | 2017-05-16 | Boston Scientific Scimed, Inc. | Bipolar off-wall electrode device for renal nerve ablation |
| US9668811B2 (en) | 2010-11-16 | 2017-06-06 | Boston Scientific Scimed, Inc. | Minimally invasive access for renal nerve ablation |
| US9687166B2 (en) | 2013-10-14 | 2017-06-27 | Boston Scientific Scimed, Inc. | High resolution cardiac mapping electrode array catheter |
| US9693821B2 (en) | 2013-03-11 | 2017-07-04 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9707036B2 (en) | 2013-06-25 | 2017-07-18 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation using localized indifferent electrodes |
| US9713730B2 (en) | 2004-09-10 | 2017-07-25 | Boston Scientific Scimed, Inc. | Apparatus and method for treatment of in-stent restenosis |
| US9770606B2 (en) | 2013-10-15 | 2017-09-26 | Boston Scientific Scimed, Inc. | Ultrasound ablation catheter with cooling infusion and centering basket |
| US9808300B2 (en) | 2006-05-02 | 2017-11-07 | Boston Scientific Scimed, Inc. | Control of arterial smooth muscle tone |
| US9808311B2 (en) | 2013-03-13 | 2017-11-07 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US9827039B2 (en) | 2013-03-15 | 2017-11-28 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9833283B2 (en) | 2013-07-01 | 2017-12-05 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US9895194B2 (en) | 2013-09-04 | 2018-02-20 | Boston Scientific Scimed, Inc. | Radio frequency (RF) balloon catheter having flushing and cooling capability |
| US9907609B2 (en) | 2014-02-04 | 2018-03-06 | Boston Scientific Scimed, Inc. | Alternative placement of thermal sensors on bipolar electrode |
| US9925001B2 (en) | 2013-07-19 | 2018-03-27 | Boston Scientific Scimed, Inc. | Spiral bipolar electrode renal denervation balloon |
| US9943365B2 (en) | 2013-06-21 | 2018-04-17 | Boston Scientific Scimed, Inc. | Renal denervation balloon catheter with ride along electrode support |
| US9956033B2 (en) | 2013-03-11 | 2018-05-01 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9962223B2 (en) | 2013-10-15 | 2018-05-08 | Boston Scientific Scimed, Inc. | Medical device balloon |
| US9974607B2 (en) | 2006-10-18 | 2018-05-22 | Vessix Vascular, Inc. | Inducing desirable temperature effects on body tissue |
| US10022182B2 (en) | 2013-06-21 | 2018-07-17 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation having rotatable shafts |
| US10085799B2 (en) | 2011-10-11 | 2018-10-02 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US10265122B2 (en) | 2013-03-15 | 2019-04-23 | Boston Scientific Scimed, Inc. | Nerve ablation devices and related methods of use |
| US10271898B2 (en) | 2013-10-25 | 2019-04-30 | Boston Scientific Scimed, Inc. | Embedded thermocouple in denervation flex circuit |
| US10321946B2 (en) | 2012-08-24 | 2019-06-18 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices with weeping RF ablation balloons |
| US10342609B2 (en) | 2013-07-22 | 2019-07-09 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US10398464B2 (en) | 2012-09-21 | 2019-09-03 | Boston Scientific Scimed, Inc. | System for nerve modulation and innocuous thermal gradient nerve block |
| US10413357B2 (en) | 2013-07-11 | 2019-09-17 | Boston Scientific Scimed, Inc. | Medical device with stretchable electrode assemblies |
| US10524859B2 (en) | 2016-06-07 | 2020-01-07 | Metavention, Inc. | Therapeutic tissue modulation devices and methods |
| US10543034B2 (en) | 2011-12-09 | 2020-01-28 | Metavention, Inc. | Modulation of nerves innervating the liver |
| US10549127B2 (en) | 2012-09-21 | 2020-02-04 | Boston Scientific Scimed, Inc. | Self-cooling ultrasound ablation catheter |
| US10660698B2 (en) | 2013-07-11 | 2020-05-26 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation |
| US10660703B2 (en) | 2012-05-08 | 2020-05-26 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices |
| US10695124B2 (en) | 2013-07-22 | 2020-06-30 | Boston Scientific Scimed, Inc. | Renal nerve ablation catheter having twist balloon |
| US10722300B2 (en) | 2013-08-22 | 2020-07-28 | Boston Scientific Scimed, Inc. | Flexible circuit having improved adhesion to a renal nerve modulation balloon |
| US10835305B2 (en) | 2012-10-10 | 2020-11-17 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices and methods |
| US10945786B2 (en) | 2013-10-18 | 2021-03-16 | Boston Scientific Scimed, Inc. | Balloon catheters with flexible conducting wires and related methods of use and manufacture |
| US10952790B2 (en) | 2013-09-13 | 2021-03-23 | Boston Scientific Scimed, Inc. | Ablation balloon with vapor deposited cover layer |
| US11000679B2 (en) | 2014-02-04 | 2021-05-11 | Boston Scientific Scimed, Inc. | Balloon protection and rewrapping devices and related methods of use |
| US11202671B2 (en) | 2014-01-06 | 2021-12-21 | Boston Scientific Scimed, Inc. | Tear resistant flex circuit assembly |
| US11246654B2 (en) | 2013-10-14 | 2022-02-15 | Boston Scientific Scimed, Inc. | Flexible renal nerve ablation devices and related methods of use and manufacture |
Families Citing this family (575)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7992572B2 (en) | 1998-06-10 | 2011-08-09 | Asthmatx, Inc. | Methods of evaluating individuals having reversible obstructive pulmonary disease |
| US6634363B1 (en) | 1997-04-07 | 2003-10-21 | Broncus Technologies, Inc. | Methods of treating lungs having reversible obstructive pulmonary disease |
| US7027869B2 (en) | 1998-01-07 | 2006-04-11 | Asthmatx, Inc. | Method for treating an asthma attack |
| US6488673B1 (en) * | 1997-04-07 | 2002-12-03 | Broncus Technologies, Inc. | Method of increasing gas exchange of a lung |
| US7921855B2 (en) | 1998-01-07 | 2011-04-12 | Asthmatx, Inc. | Method for treating an asthma attack |
| US7198635B2 (en) | 2000-10-17 | 2007-04-03 | Asthmatx, Inc. | Modification of airways by application of energy |
| US8181656B2 (en) | 1998-06-10 | 2012-05-22 | Asthmatx, Inc. | Methods for treating airways |
| US7137980B2 (en) | 1998-10-23 | 2006-11-21 | Sherwood Services Ag | Method and system for controlling output of RF medical generator |
| US8251070B2 (en) | 2000-03-27 | 2012-08-28 | Asthmatx, Inc. | Methods for treating airways |
| CA2416581A1 (en) | 2000-07-25 | 2002-04-25 | Rita Medical Systems, Inc. | Apparatus for detecting and treating tumors using localized impedance measurement |
| US7104987B2 (en) | 2000-10-17 | 2006-09-12 | Asthmatx, Inc. | Control system and process for application of energy to airway walls and other mediums |
| US6994706B2 (en) | 2001-08-13 | 2006-02-07 | Minnesota Medical Physics, Llc | Apparatus and method for treatment of benign prostatic hyperplasia |
| US20050033137A1 (en) * | 2002-10-25 | 2005-02-10 | The Regents Of The University Of Michigan | Ablation catheters and methods for their use |
| US20040082947A1 (en) | 2002-10-25 | 2004-04-29 | The Regents Of The University Of Michigan | Ablation catheters |
| US7255694B2 (en) * | 2002-12-10 | 2007-08-14 | Sherwood Services Ag | Variable output crest factor electrosurgical generator |
| US20040226556A1 (en) | 2003-05-13 | 2004-11-18 | Deem Mark E. | Apparatus for treating asthma using neurotoxin |
| US9060770B2 (en) | 2003-05-20 | 2015-06-23 | Ethicon Endo-Surgery, Inc. | Robotically-driven surgical instrument with E-beam driver |
| US20070084897A1 (en) | 2003-05-20 | 2007-04-19 | Shelton Frederick E Iv | Articulating surgical stapling instrument incorporating a two-piece e-beam firing mechanism |
| GB2403148C2 (en) | 2003-06-23 | 2013-02-13 | Microsulis Ltd | Radiation applicator |
| US7396336B2 (en) | 2003-10-30 | 2008-07-08 | Sherwood Services Ag | Switched resonant ultrasonic power amplifier system |
| US8409219B2 (en) | 2004-06-18 | 2013-04-02 | Medtronic, Inc. | Method and system for placement of electrical lead inside heart |
| GB2415630C2 (en) | 2004-07-02 | 2007-03-22 | Microsulis Ltd | Radiation applicator and method of radiating tissue |
| US11896225B2 (en) | 2004-07-28 | 2024-02-13 | Cilag Gmbh International | Staple cartridge comprising a pan |
| US8215531B2 (en) | 2004-07-28 | 2012-07-10 | Ethicon Endo-Surgery, Inc. | Surgical stapling instrument having a medical substance dispenser |
| US8182433B2 (en) * | 2005-03-04 | 2012-05-22 | Endosense Sa | Medical apparatus system having optical fiber load sensing capability |
| US8075498B2 (en) | 2005-03-04 | 2011-12-13 | Endosense Sa | Medical apparatus system having optical fiber load sensing capability |
| US9474564B2 (en) | 2005-03-31 | 2016-10-25 | Covidien Ag | Method and system for compensating for external impedance of an energy carrying component when controlling an electrosurgical generator |
| CA2605360C (en) * | 2005-04-21 | 2017-03-28 | Asthmatx, Inc. | Control methods and devices for energy delivery |
| GB2434314B (en) | 2006-01-03 | 2011-06-15 | Microsulis Ltd | Microwave applicator with dipole antenna |
| EP2363073B1 (en) | 2005-08-01 | 2015-10-07 | St. Jude Medical Luxembourg Holding S.à.r.l. | Medical apparatus system having optical fiber load sensing capability |
| US9237891B2 (en) | 2005-08-31 | 2016-01-19 | Ethicon Endo-Surgery, Inc. | Robotically-controlled surgical stapling devices that produce formed staples having different lengths |
| US7669746B2 (en) | 2005-08-31 | 2010-03-02 | Ethicon Endo-Surgery, Inc. | Staple cartridges for forming staples having differing formed staple heights |
| US7934630B2 (en) | 2005-08-31 | 2011-05-03 | Ethicon Endo-Surgery, Inc. | Staple cartridges for forming staples having differing formed staple heights |
| US10159482B2 (en) | 2005-08-31 | 2018-12-25 | Ethicon Llc | Fastener cartridge assembly comprising a fixed anvil and different staple heights |
| US11246590B2 (en) | 2005-08-31 | 2022-02-15 | Cilag Gmbh International | Staple cartridge including staple drivers having different unfired heights |
| US8317070B2 (en) | 2005-08-31 | 2012-11-27 | Ethicon Endo-Surgery, Inc. | Surgical stapling devices that produce formed staples having different lengths |
| US11484312B2 (en) | 2005-08-31 | 2022-11-01 | Cilag Gmbh International | Staple cartridge comprising a staple driver arrangement |
| US8734438B2 (en) | 2005-10-21 | 2014-05-27 | Covidien Ag | Circuit and method for reducing stored energy in an electrosurgical generator |
| US20070106317A1 (en) | 2005-11-09 | 2007-05-10 | Shelton Frederick E Iv | Hydraulically and electrically actuated articulation joints for surgical instruments |
| CN101505674B (en) | 2006-01-17 | 2014-03-05 | 恩迪梅德医疗有限公司 | Electrosurgical methods and devices employing phase-controlled radiofrequency energy |
| US9186200B2 (en) | 2006-01-24 | 2015-11-17 | Covidien Ag | System and method for tissue sealing |
| CA2574935A1 (en) | 2006-01-24 | 2007-07-24 | Sherwood Services Ag | A method and system for controlling an output of a radio-frequency medical generator having an impedance based control algorithm |
| US8186555B2 (en) | 2006-01-31 | 2012-05-29 | Ethicon Endo-Surgery, Inc. | Motor-driven surgical cutting and fastening instrument with mechanical closure system |
| US20110024477A1 (en) | 2009-02-06 | 2011-02-03 | Hall Steven G | Driven Surgical Stapler Improvements |
| US8820603B2 (en) | 2006-01-31 | 2014-09-02 | Ethicon Endo-Surgery, Inc. | Accessing data stored in a memory of a surgical instrument |
| US7753904B2 (en) | 2006-01-31 | 2010-07-13 | Ethicon Endo-Surgery, Inc. | Endoscopic surgical instrument with a handle that can articulate with respect to the shaft |
| US11278279B2 (en) | 2006-01-31 | 2022-03-22 | Cilag Gmbh International | Surgical instrument assembly |
| US11793518B2 (en) | 2006-01-31 | 2023-10-24 | Cilag Gmbh International | Powered surgical instruments with firing system lockout arrangements |
| US11224427B2 (en) | 2006-01-31 | 2022-01-18 | Cilag Gmbh International | Surgical stapling system including a console and retraction assembly |
| US20120292367A1 (en) | 2006-01-31 | 2012-11-22 | Ethicon Endo-Surgery, Inc. | Robotically-controlled end effector |
| US20110006101A1 (en) | 2009-02-06 | 2011-01-13 | EthiconEndo-Surgery, Inc. | Motor driven surgical fastener device with cutting member lockout arrangements |
| US9861359B2 (en) | 2006-01-31 | 2018-01-09 | Ethicon Llc | Powered surgical instruments with firing system lockout arrangements |
| US8708213B2 (en) | 2006-01-31 | 2014-04-29 | Ethicon Endo-Surgery, Inc. | Surgical instrument having a feedback system |
| US7845537B2 (en) | 2006-01-31 | 2010-12-07 | Ethicon Endo-Surgery, Inc. | Surgical instrument having recording capabilities |
| US20110295295A1 (en) | 2006-01-31 | 2011-12-01 | Ethicon Endo-Surgery, Inc. | Robotically-controlled surgical instrument having recording capabilities |
| US8992422B2 (en) | 2006-03-23 | 2015-03-31 | Ethicon Endo-Surgery, Inc. | Robotically-controlled endoscopic accessory channel |
| US20070225562A1 (en) | 2006-03-23 | 2007-09-27 | Ethicon Endo-Surgery, Inc. | Articulating endoscopic accessory channel |
| US7651492B2 (en) | 2006-04-24 | 2010-01-26 | Covidien Ag | Arc based adaptive control system for an electrosurgical unit |
| US8567265B2 (en) | 2006-06-09 | 2013-10-29 | Endosense, SA | Triaxial fiber optic force sensing catheter |
| US8048063B2 (en) | 2006-06-09 | 2011-11-01 | Endosense Sa | Catheter having tri-axial force sensor |
| US8322455B2 (en) | 2006-06-27 | 2012-12-04 | Ethicon Endo-Surgery, Inc. | Manually driven surgical cutting and fastening instrument |
| ES2928065T3 (en) * | 2006-06-28 | 2022-11-15 | Medtronic Ardian Luxembourg | Thermally induced renal neuromodulation systems |
| US7722603B2 (en) * | 2006-09-28 | 2010-05-25 | Covidien Ag | Smart return electrode pad |
| US8220690B2 (en) | 2006-09-29 | 2012-07-17 | Ethicon Endo-Surgery, Inc. | Connected surgical staples and stapling instruments for deploying the same |
| US10568652B2 (en) | 2006-09-29 | 2020-02-25 | Ethicon Llc | Surgical staples having attached drivers of different heights and stapling instruments for deploying the same |
| US8684253B2 (en) | 2007-01-10 | 2014-04-01 | Ethicon Endo-Surgery, Inc. | Surgical instrument with wireless communication between a control unit of a robotic system and remote sensor |
| US8652120B2 (en) | 2007-01-10 | 2014-02-18 | Ethicon Endo-Surgery, Inc. | Surgical instrument with wireless communication between control unit and sensor transponders |
| US11291441B2 (en) | 2007-01-10 | 2022-04-05 | Cilag Gmbh International | Surgical instrument with wireless communication between control unit and remote sensor |
| US8827133B2 (en) | 2007-01-11 | 2014-09-09 | Ethicon Endo-Surgery, Inc. | Surgical stapling device having supports for a flexible drive mechanism |
| US11039836B2 (en) | 2007-01-11 | 2021-06-22 | Cilag Gmbh International | Staple cartridge for use with a surgical stapling instrument |
| WO2008125962A2 (en) * | 2007-03-01 | 2008-10-23 | Endymed Medical Ltd. | Electrosurgical methods and devices employing semiconductor chips |
| WO2008118737A1 (en) * | 2007-03-22 | 2008-10-02 | University Of Virginia Patent Foundation | Electrode catheter for ablation purposes and related method thereof |
| US10166066B2 (en) * | 2007-03-13 | 2019-01-01 | University Of Virginia Patent Foundation | Epicardial ablation catheter and method of use |
| US7669747B2 (en) | 2007-03-15 | 2010-03-02 | Ethicon Endo-Surgery, Inc. | Washer for use with a surgical stapling instrument |
| US9468396B2 (en) | 2007-03-19 | 2016-10-18 | University Of Virginia Patent Foundation | Systems and methods for determining location of an access needle in a subject |
| US11058354B2 (en) | 2007-03-19 | 2021-07-13 | University Of Virginia Patent Foundation | Access needle with direct visualization and related methods |
| WO2008115745A2 (en) | 2007-03-19 | 2008-09-25 | University Of Virginia Patent Foundation | Access needle pressure sensor device and method of use |
| US8893946B2 (en) | 2007-03-28 | 2014-11-25 | Ethicon Endo-Surgery, Inc. | Laparoscopic tissue thickness and clamp load measuring devices |
| US8157789B2 (en) * | 2007-05-24 | 2012-04-17 | Endosense Sa | Touch sensing catheter |
| US8622935B1 (en) | 2007-05-25 | 2014-01-07 | Endosense Sa | Elongated surgical manipulator with body position and distal force sensing |
| US11857181B2 (en) | 2007-06-04 | 2024-01-02 | Cilag Gmbh International | Robotically-controlled shaft based rotary drive systems for surgical instruments |
| US8931682B2 (en) | 2007-06-04 | 2015-01-13 | Ethicon Endo-Surgery, Inc. | Robotically-controlled shaft based rotary drive systems for surgical instruments |
| US7753245B2 (en) | 2007-06-22 | 2010-07-13 | Ethicon Endo-Surgery, Inc. | Surgical stapling instruments |
| US8408439B2 (en) | 2007-06-22 | 2013-04-02 | Ethicon Endo-Surgery, Inc. | Surgical stapling instrument with an articulatable end effector |
| US11849941B2 (en) | 2007-06-29 | 2023-12-26 | Cilag Gmbh International | Staple cartridge having staple cavities extending at a transverse angle relative to a longitudinal cartridge axis |
| JP5436423B2 (en) | 2007-07-24 | 2014-03-05 | アスマティックス,インコーポレイテッド | System and method for power control based on impedance detection such as power control for tissue treatment device |
| US8512332B2 (en) | 2007-09-21 | 2013-08-20 | Covidien Lp | Real-time arc control in electrosurgical generators |
| US20100241185A1 (en) * | 2007-11-09 | 2010-09-23 | University Of Virginia Patent Foundation | Steerable epicardial pacing catheter system placed via the subxiphoid process |
| US8906011B2 (en) | 2007-11-16 | 2014-12-09 | Kardium Inc. | Medical device for use in bodily lumens, for example an atrium |
| US8758391B2 (en) | 2008-02-14 | 2014-06-24 | Ethicon Endo-Surgery, Inc. | Interchangeable tools for surgical instruments |
| US7866527B2 (en) | 2008-02-14 | 2011-01-11 | Ethicon Endo-Surgery, Inc. | Surgical stapling apparatus with interlockable firing system |
| BRPI0901282A2 (en) | 2008-02-14 | 2009-11-17 | Ethicon Endo Surgery Inc | surgical cutting and fixation instrument with rf electrodes |
| US8657174B2 (en) * | 2008-02-14 | 2014-02-25 | Ethicon Endo-Surgery, Inc. | Motorized surgical cutting and fastening instrument having handle based power source |
| US8573465B2 (en) | 2008-02-14 | 2013-11-05 | Ethicon Endo-Surgery, Inc. | Robotically-controlled surgical end effector system with rotary actuated closure systems |
| US7819298B2 (en) | 2008-02-14 | 2010-10-26 | Ethicon Endo-Surgery, Inc. | Surgical stapling apparatus with control features operable with one hand |
| US9179912B2 (en) | 2008-02-14 | 2015-11-10 | Ethicon Endo-Surgery, Inc. | Robotically-controlled motorized surgical cutting and fastening instrument |
| US8636736B2 (en) | 2008-02-14 | 2014-01-28 | Ethicon Endo-Surgery, Inc. | Motorized surgical cutting and fastening instrument |
| US8483831B1 (en) | 2008-02-15 | 2013-07-09 | Holaira, Inc. | System and method for bronchial dilation |
| US11272927B2 (en) | 2008-02-15 | 2022-03-15 | Cilag Gmbh International | Layer arrangements for surgical staple cartridges |
| US9770245B2 (en) | 2008-02-15 | 2017-09-26 | Ethicon Llc | Layer arrangements for surgical staple cartridges |
| US9867652B2 (en) | 2008-04-29 | 2018-01-16 | Virginia Tech Intellectual Properties, Inc. | Irreversible electroporation using tissue vasculature to treat aberrant cell masses or create tissue scaffolds |
| US8992517B2 (en) | 2008-04-29 | 2015-03-31 | Virginia Tech Intellectual Properties Inc. | Irreversible electroporation to treat aberrant cell masses |
| US9198733B2 (en) | 2008-04-29 | 2015-12-01 | Virginia Tech Intellectual Properties, Inc. | Treatment planning for electroporation-based therapies |
| US11254926B2 (en) | 2008-04-29 | 2022-02-22 | Virginia Tech Intellectual Properties, Inc. | Devices and methods for high frequency electroporation |
| EP2280741A4 (en) | 2008-04-29 | 2012-06-13 | Virginia Tech Intell Prop | Irreversible electroporation to create tissue scaffolds |
| US9283051B2 (en) | 2008-04-29 | 2016-03-15 | Virginia Tech Intellectual Properties, Inc. | System and method for estimating a treatment volume for administering electrical-energy based therapies |
| US11272979B2 (en) | 2008-04-29 | 2022-03-15 | Virginia Tech Intellectual Properties, Inc. | System and method for estimating tissue heating of a target ablation zone for electrical-energy based therapies |
| US10245098B2 (en) | 2008-04-29 | 2019-04-02 | Virginia Tech Intellectual Properties, Inc. | Acute blood-brain barrier disruption using electrical energy based therapy |
| US10272178B2 (en) | 2008-04-29 | 2019-04-30 | Virginia Tech Intellectual Properties Inc. | Methods for blood-brain barrier disruption using electrical energy |
| US10238447B2 (en) | 2008-04-29 | 2019-03-26 | Virginia Tech Intellectual Properties, Inc. | System and method for ablating a tissue site by electroporation with real-time monitoring of treatment progress |
| US10117707B2 (en) | 2008-04-29 | 2018-11-06 | Virginia Tech Intellectual Properties, Inc. | System and method for estimating tissue heating of a target ablation zone for electrical-energy based therapies |
| US10702326B2 (en) | 2011-07-15 | 2020-07-07 | Virginia Tech Intellectual Properties, Inc. | Device and method for electroporation based treatment of stenosis of a tubular body part |
| ES2398052T5 (en) | 2008-05-09 | 2021-10-25 | Nuvaira Inc | Systems for treating a bronchial tree |
| US8298227B2 (en) * | 2008-05-14 | 2012-10-30 | Endosense Sa | Temperature compensated strain sensing catheter |
| US9005230B2 (en) | 2008-09-23 | 2015-04-14 | Ethicon Endo-Surgery, Inc. | Motorized surgical instrument |
| US11648005B2 (en) | 2008-09-23 | 2023-05-16 | Cilag Gmbh International | Robotically-controlled motorized surgical instrument with an end effector |
| US8210411B2 (en) | 2008-09-23 | 2012-07-03 | Ethicon Endo-Surgery, Inc. | Motor-driven surgical cutting instrument |
| US9386983B2 (en) | 2008-09-23 | 2016-07-12 | Ethicon Endo-Surgery, Llc | Robotically-controlled motorized surgical instrument |
| US8608045B2 (en) | 2008-10-10 | 2013-12-17 | Ethicon Endo-Sugery, Inc. | Powered surgical cutting and stapling apparatus with manually retractable firing system |
| US9795442B2 (en) | 2008-11-11 | 2017-10-24 | Shifamed Holdings, Llc | Ablation catheters |
| US8517239B2 (en) | 2009-02-05 | 2013-08-27 | Ethicon Endo-Surgery, Inc. | Surgical stapling instrument comprising a magnetic element driver |
| CA2751664A1 (en) | 2009-02-06 | 2010-08-12 | Ethicon Endo-Surgery, Inc. | Driven surgical stapler improvements |
| US8444036B2 (en) | 2009-02-06 | 2013-05-21 | Ethicon Endo-Surgery, Inc. | Motor driven surgical fastener device with mechanisms for adjusting a tissue gap within the end effector |
| US10166067B2 (en) * | 2009-02-12 | 2019-01-01 | St. Jude Medical, Atrial Fibrillation Division, Inc. | Ablation catheter and method for electically isolating cardiac tissue |
| US11638603B2 (en) | 2009-04-09 | 2023-05-02 | Virginia Tech Intellectual Properties, Inc. | Selective modulation of intracellular effects of cells using pulsed electric fields |
| US11382681B2 (en) | 2009-04-09 | 2022-07-12 | Virginia Tech Intellectual Properties, Inc. | Device and methods for delivery of high frequency electrical pulses for non-thermal ablation |
| US8903488B2 (en) | 2009-05-28 | 2014-12-02 | Angiodynamics, Inc. | System and method for synchronizing energy delivery to the cardiac rhythm |
| US9895189B2 (en) | 2009-06-19 | 2018-02-20 | Angiodynamics, Inc. | Methods of sterilization and treating infection using irreversible electroporation |
| US8323241B2 (en) * | 2009-06-24 | 2012-12-04 | Shifamed Holdings, Llc | Steerable medical delivery devices and methods of use |
| US8932282B2 (en) | 2009-08-03 | 2015-01-13 | Covidien Lp | Power level transitioning in a surgical instrument |
| US9572624B2 (en) | 2009-08-05 | 2017-02-21 | Atricure, Inc. | Bipolar belt systems and methods |
| US9642534B2 (en) | 2009-09-11 | 2017-05-09 | University Of Virginia Patent Foundation | Systems and methods for determining location of an access needle in a subject |
| GB2474233A (en) | 2009-10-06 | 2011-04-13 | Uk Investments Associates Llc | Cooling pump comprising a detachable head portion |
| EP2493408B1 (en) | 2009-10-27 | 2015-06-24 | Holaira, Inc. | Delivery devices with coolable energy emitting assemblies |
| KR101820542B1 (en) | 2009-11-11 | 2018-01-19 | 호라이라 인코포레이티드 | Systems, apparatuses, and methods for treating tissue and controlling stenosis |
| US8911439B2 (en) | 2009-11-11 | 2014-12-16 | Holaira, Inc. | Non-invasive and minimally invasive denervation methods and systems for performing the same |
| US8851354B2 (en) | 2009-12-24 | 2014-10-07 | Ethicon Endo-Surgery, Inc. | Surgical cutting instrument that analyzes tissue thickness |
| US8220688B2 (en) | 2009-12-24 | 2012-07-17 | Ethicon Endo-Surgery, Inc. | Motor-driven surgical cutting instrument with electric actuator directional control assembly |
| EP2537149B1 (en) | 2010-02-18 | 2017-10-25 | University Of Virginia Patent Foundation | System, method, and computer program product for simulating epicardial electrophysiology procedures |
| JP6045916B2 (en) * | 2010-02-26 | 2016-12-14 | コーニンクレッカ フィリップス エヌ ヴェKoninklijke Philips N.V. | Interventional ablation device with tissue identification capability |
| CN102917748B (en) | 2010-03-24 | 2015-04-01 | 施菲姆德控股有限责任公司 | Intravascular tissue disruption |
| US9655677B2 (en) | 2010-05-12 | 2017-05-23 | Shifamed Holdings, Llc | Ablation catheters including a balloon and electrodes |
| AU2011252976A1 (en) | 2010-05-12 | 2012-11-08 | Shifamed Holdings, Llc | Low profile electrode assembly |
| US8783543B2 (en) | 2010-07-30 | 2014-07-22 | Ethicon Endo-Surgery, Inc. | Tissue acquisition arrangements and methods for surgical stapling devices |
| US8632525B2 (en) | 2010-09-17 | 2014-01-21 | Ethicon Endo-Surgery, Inc. | Power control arrangements for surgical instruments and batteries |
| US9289212B2 (en) | 2010-09-17 | 2016-03-22 | Ethicon Endo-Surgery, Inc. | Surgical instruments and batteries for surgical instruments |
| US9320523B2 (en) | 2012-03-28 | 2016-04-26 | Ethicon Endo-Surgery, Llc | Tissue thickness compensator comprising tissue ingrowth features |
| US11849952B2 (en) | 2010-09-30 | 2023-12-26 | Cilag Gmbh International | Staple cartridge comprising staples positioned within a compressible portion thereof |
| US9351730B2 (en) | 2011-04-29 | 2016-05-31 | Ethicon Endo-Surgery, Llc | Tissue thickness compensator comprising channels |
| US9517063B2 (en) | 2012-03-28 | 2016-12-13 | Ethicon Endo-Surgery, Llc | Movable member for use with a tissue thickness compensator |
| US10945731B2 (en) | 2010-09-30 | 2021-03-16 | Ethicon Llc | Tissue thickness compensator comprising controlled release and expansion |
| BR112013007717B1 (en) | 2010-09-30 | 2020-09-24 | Ethicon Endo-Surgery, Inc. | SURGICAL CLAMPING SYSTEM |
| US9629814B2 (en) | 2010-09-30 | 2017-04-25 | Ethicon Endo-Surgery, Llc | Tissue thickness compensator configured to redistribute compressive forces |
| US8978954B2 (en) | 2010-09-30 | 2015-03-17 | Ethicon Endo-Surgery, Inc. | Staple cartridge comprising an adjustable distal portion |
| US9301753B2 (en) | 2010-09-30 | 2016-04-05 | Ethicon Endo-Surgery, Llc | Expandable tissue thickness compensator |
| US9592050B2 (en) | 2010-09-30 | 2017-03-14 | Ethicon Endo-Surgery, Llc | End effector comprising a distal tissue abutment member |
| US11812965B2 (en) | 2010-09-30 | 2023-11-14 | Cilag Gmbh International | Layer of material for a surgical end effector |
| US11298125B2 (en) | 2010-09-30 | 2022-04-12 | Cilag Gmbh International | Tissue stapler having a thickness compensator |
| US9220501B2 (en) | 2010-09-30 | 2015-12-29 | Ethicon Endo-Surgery, Inc. | Tissue thickness compensators |
| US9364233B2 (en) | 2010-09-30 | 2016-06-14 | Ethicon Endo-Surgery, Llc | Tissue thickness compensators for circular surgical staplers |
| US9220500B2 (en) | 2010-09-30 | 2015-12-29 | Ethicon Endo-Surgery, Inc. | Tissue thickness compensator comprising structure to produce a resilient load |
| US9216019B2 (en) | 2011-09-23 | 2015-12-22 | Ethicon Endo-Surgery, Inc. | Surgical stapler with stationary staple drivers |
| US8695866B2 (en) | 2010-10-01 | 2014-04-15 | Ethicon Endo-Surgery, Inc. | Surgical instrument having a power control circuit |
| WO2012051433A2 (en) | 2010-10-13 | 2012-04-19 | Angiodynamics, Inc. | System and method for electrically ablating tissue of a patient |
| CN107007348B (en) | 2010-10-25 | 2019-05-31 | 美敦力Af卢森堡有限责任公司 | For the estimation of nerve modulation treatment and device, the system and method for feedback |
| US9532828B2 (en) | 2010-11-29 | 2017-01-03 | Medtronic Ablation Frontiers Llc | System and method for adaptive RF ablation |
| US9737353B2 (en) | 2010-12-16 | 2017-08-22 | Biosense Webster (Israel) Ltd. | System for controlling tissue ablation using temperature sensors |
| WO2012088149A2 (en) | 2010-12-20 | 2012-06-28 | Virginia Tech Intellectual Properties, Inc. | High-frequency electroporation for cancer therapy |
| US9044245B2 (en) * | 2011-01-05 | 2015-06-02 | Medtronic Ablation Frontiers Llc | Multipolarity epicardial radiofrequency ablation |
| CA2764494A1 (en) | 2011-01-21 | 2012-07-21 | Kardium Inc. | Enhanced medical device for use in bodily cavities, for example an atrium |
| US11259867B2 (en) | 2011-01-21 | 2022-03-01 | Kardium Inc. | High-density electrode-based medical device system |
| US9486273B2 (en) | 2011-01-21 | 2016-11-08 | Kardium Inc. | High-density electrode-based medical device system |
| US9452016B2 (en) | 2011-01-21 | 2016-09-27 | Kardium Inc. | Catheter system |
| JP6177230B2 (en) | 2011-04-14 | 2017-08-09 | セント・ジュード・メディカル・インターナショナル・ホールディング・エスエーアールエルSt. Jude Medical International Holding S.a,r.l. | Small force sensor for catheter |
| CA2834649C (en) | 2011-04-29 | 2021-02-16 | Ethicon Endo-Surgery, Inc. | Staple cartridge comprising staples positioned within a compressible portion thereof |
| AU2012250729B2 (en) | 2011-05-03 | 2016-12-01 | Shifamed Holdings, Llc | Steerable delivery sheaths |
| US9072535B2 (en) | 2011-05-27 | 2015-07-07 | Ethicon Endo-Surgery, Inc. | Surgical stapling instruments with rotatable staple deployment arrangements |
| US11207064B2 (en) | 2011-05-27 | 2021-12-28 | Cilag Gmbh International | Automated end effector component reloading system for use with a robotic system |
| US9277960B2 (en) | 2011-09-08 | 2016-03-08 | Kardium Inc. | Intra-cardiac mapping and ablating |
| US9050084B2 (en) | 2011-09-23 | 2015-06-09 | Ethicon Endo-Surgery, Inc. | Staple cartridge including collapsible deck arrangement |
| US9078665B2 (en) | 2011-09-28 | 2015-07-14 | Angiodynamics, Inc. | Multiple treatment zone ablation probe |
| CA2852414C (en) * | 2011-10-15 | 2020-12-22 | Diros Technology Inc. | Method and apparatus for precisely controlling the size and shape of radiofrequency ablations |
| US9044230B2 (en) | 2012-02-13 | 2015-06-02 | Ethicon Endo-Surgery, Inc. | Surgical cutting and fastening instrument with apparatus for determining cartridge and firing motion status |
| JP6224070B2 (en) | 2012-03-28 | 2017-11-01 | エシコン・エンド−サージェリィ・インコーポレイテッドEthicon Endo−Surgery,Inc. | Retainer assembly including tissue thickness compensator |
| CN104334098B (en) | 2012-03-28 | 2017-03-22 | 伊西康内外科公司 | Tissue thickness compensator comprising capsules defining a low pressure environment |
| JP6305979B2 (en) | 2012-03-28 | 2018-04-04 | エシコン・エンド−サージェリィ・インコーポレイテッドEthicon Endo−Surgery,Inc. | Tissue thickness compensator with multiple layers |
| US8961550B2 (en) | 2012-04-17 | 2015-02-24 | Indian Wells Medical, Inc. | Steerable endoluminal punch |
| US9770293B2 (en) | 2012-06-04 | 2017-09-26 | Boston Scientific Scimed, Inc. | Systems and methods for treating tissue of a passageway within a body |
| US9101358B2 (en) | 2012-06-15 | 2015-08-11 | Ethicon Endo-Surgery, Inc. | Articulatable surgical instrument comprising a firing drive |
| US20140005718A1 (en) | 2012-06-28 | 2014-01-02 | Ethicon Endo-Surgery, Inc. | Multi-functional powered surgical device with external dissection features |
| CN104487005B (en) | 2012-06-28 | 2017-09-08 | 伊西康内外科公司 | Empty squeeze latching member |
| US20140001231A1 (en) | 2012-06-28 | 2014-01-02 | Ethicon Endo-Surgery, Inc. | Firing system lockout arrangements for surgical instruments |
| US9649111B2 (en) | 2012-06-28 | 2017-05-16 | Ethicon Endo-Surgery, Llc | Replaceable clip cartridge for a clip applier |
| BR112014032776B1 (en) | 2012-06-28 | 2021-09-08 | Ethicon Endo-Surgery, Inc | SURGICAL INSTRUMENT SYSTEM AND SURGICAL KIT FOR USE WITH A SURGICAL INSTRUMENT SYSTEM |
| US9289256B2 (en) | 2012-06-28 | 2016-03-22 | Ethicon Endo-Surgery, Llc | Surgical end effectors having angled tissue-contacting surfaces |
| US11278284B2 (en) | 2012-06-28 | 2022-03-22 | Cilag Gmbh International | Rotary drive arrangements for surgical instruments |
| US20140001234A1 (en) | 2012-06-28 | 2014-01-02 | Ethicon Endo-Surgery, Inc. | Coupling arrangements for attaching surgical end effectors to drive systems therefor |
| US9529025B2 (en) | 2012-06-29 | 2016-12-27 | Covidien Lp | Systems and methods for measuring the frequency of signals generated by high frequency medical devices |
| EP2877113B1 (en) | 2012-07-24 | 2018-07-25 | Boston Scientific Scimed, Inc. | Electrodes for tissue treatment |
| US9272132B2 (en) | 2012-11-02 | 2016-03-01 | Boston Scientific Scimed, Inc. | Medical device for treating airways and related methods of use |
| WO2014071372A1 (en) | 2012-11-05 | 2014-05-08 | Boston Scientific Scimed, Inc. | Devices for delivering energy to body lumens |
| US9398933B2 (en) | 2012-12-27 | 2016-07-26 | Holaira, Inc. | Methods for improving drug efficacy including a combination of drug administration and nerve modulation |
| US20140200639A1 (en) * | 2013-01-16 | 2014-07-17 | Advanced Neuromodulation Systems, Inc. | Self-expanding neurostimulation leads having broad multi-electrode arrays |
| MX368026B (en) | 2013-03-01 | 2019-09-12 | Ethicon Endo Surgery Inc | Articulatable surgical instruments with conductive pathways for signal communication. |
| US9700309B2 (en) | 2013-03-01 | 2017-07-11 | Ethicon Llc | Articulatable surgical instruments with conductive pathways for signal communication |
| MX364729B (en) | 2013-03-01 | 2019-05-06 | Ethicon Endo Surgery Inc | Surgical instrument with a soft stop. |
| US20140263552A1 (en) | 2013-03-13 | 2014-09-18 | Ethicon Endo-Surgery, Inc. | Staple cartridge tissue thickness sensor system |
| US9629623B2 (en) | 2013-03-14 | 2017-04-25 | Ethicon Endo-Surgery, Llc | Drive system lockout arrangements for modular surgical instruments |
| US9629629B2 (en) | 2013-03-14 | 2017-04-25 | Ethicon Endo-Surgey, LLC | Control systems for surgical instruments |
| US9795384B2 (en) | 2013-03-27 | 2017-10-24 | Ethicon Llc | Fastener cartridge comprising a tissue thickness compensator and a gap setting element |
| US9572577B2 (en) | 2013-03-27 | 2017-02-21 | Ethicon Endo-Surgery, Llc | Fastener cartridge comprising a tissue thickness compensator including openings therein |
| US10098694B2 (en) | 2013-04-08 | 2018-10-16 | Apama Medical, Inc. | Tissue ablation and monitoring thereof |
| US10349824B2 (en) | 2013-04-08 | 2019-07-16 | Apama Medical, Inc. | Tissue mapping and visualization systems |
| JP6463731B2 (en) | 2013-04-08 | 2019-02-06 | アパマ・メディカル・インコーポレーテッド | Imaging system |
| BR112015026109B1 (en) | 2013-04-16 | 2022-02-22 | Ethicon Endo-Surgery, Inc | surgical instrument |
| US10405857B2 (en) | 2013-04-16 | 2019-09-10 | Ethicon Llc | Powered linear surgical stapler |
| US9574644B2 (en) | 2013-05-30 | 2017-02-21 | Ethicon Endo-Surgery, Llc | Power module for use with a surgical instrument |
| US9872719B2 (en) | 2013-07-24 | 2018-01-23 | Covidien Lp | Systems and methods for generating electrosurgical energy using a multistage power converter |
| US9655670B2 (en) | 2013-07-29 | 2017-05-23 | Covidien Lp | Systems and methods for measuring tissue impedance through an electrosurgical cable |
| EP3030182B1 (en) | 2013-08-09 | 2018-01-10 | Boston Scientific Scimed, Inc. | Expandable catheter |
| CN106028966B (en) | 2013-08-23 | 2018-06-22 | 伊西康内外科有限责任公司 | For the firing member restoring device of powered surgical instrument |
| US9808249B2 (en) | 2013-08-23 | 2017-11-07 | Ethicon Llc | Attachment portions for surgical instrument assemblies |
| US10448986B2 (en) * | 2013-09-27 | 2019-10-22 | Covidien Lp | Electrosurgical medical device with power modulation |
| US10433902B2 (en) | 2013-10-23 | 2019-10-08 | Medtronic Ardian Luxembourg S.A.R.L. | Current control methods and systems |
| US10098685B2 (en) * | 2013-10-30 | 2018-10-16 | Medtronic Cryocath Lp | Feedback system for cryoablation of cardiac tissue |
| US9839428B2 (en) | 2013-12-23 | 2017-12-12 | Ethicon Llc | Surgical cutting and stapling instruments with independent jaw control features |
| US20150173756A1 (en) | 2013-12-23 | 2015-06-25 | Ethicon Endo-Surgery, Inc. | Surgical cutting and stapling methods |
| US9724092B2 (en) | 2013-12-23 | 2017-08-08 | Ethicon Llc | Modular surgical instruments |
| US9687232B2 (en) | 2013-12-23 | 2017-06-27 | Ethicon Llc | Surgical staples |
| US9962161B2 (en) | 2014-02-12 | 2018-05-08 | Ethicon Llc | Deliverable surgical instrument |
| US20140166726A1 (en) | 2014-02-24 | 2014-06-19 | Ethicon Endo-Surgery, Inc. | Staple cartridge including a barbed staple |
| JP6462004B2 (en) | 2014-02-24 | 2019-01-30 | エシコン エルエルシー | Fastening system with launcher lockout |
| US9820738B2 (en) | 2014-03-26 | 2017-11-21 | Ethicon Llc | Surgical instrument comprising interactive systems |
| US9913642B2 (en) | 2014-03-26 | 2018-03-13 | Ethicon Llc | Surgical instrument comprising a sensor system |
| US9750499B2 (en) | 2014-03-26 | 2017-09-05 | Ethicon Llc | Surgical stapling instrument system |
| BR112016021943B1 (en) | 2014-03-26 | 2022-06-14 | Ethicon Endo-Surgery, Llc | SURGICAL INSTRUMENT FOR USE BY AN OPERATOR IN A SURGICAL PROCEDURE |
| US9733663B2 (en) | 2014-03-26 | 2017-08-15 | Ethicon Llc | Power management through segmented circuit and variable voltage protection |
| BR112016023825B1 (en) | 2014-04-16 | 2022-08-02 | Ethicon Endo-Surgery, Llc | STAPLE CARTRIDGE FOR USE WITH A SURGICAL STAPLER AND STAPLE CARTRIDGE FOR USE WITH A SURGICAL INSTRUMENT |
| US10299792B2 (en) | 2014-04-16 | 2019-05-28 | Ethicon Llc | Fastener cartridge comprising non-uniform fasteners |
| US9801627B2 (en) | 2014-09-26 | 2017-10-31 | Ethicon Llc | Fastener cartridge for creating a flexible staple line |
| CN106456176B (en) | 2014-04-16 | 2019-06-28 | 伊西康内外科有限责任公司 | Fastener cartridge including the extension with various configuration |
| BR112016023807B1 (en) | 2014-04-16 | 2022-07-12 | Ethicon Endo-Surgery, Llc | CARTRIDGE SET OF FASTENERS FOR USE WITH A SURGICAL INSTRUMENT |
| US20150297225A1 (en) | 2014-04-16 | 2015-10-22 | Ethicon Endo-Surgery, Inc. | Fastener cartridges including extensions having different configurations |
| US10610292B2 (en) | 2014-04-25 | 2020-04-07 | Medtronic Ardian Luxembourg S.A.R.L. | Devices, systems, and methods for monitoring and/or controlling deployment of a neuromodulation element within a body lumen and related technology |
| WO2015175570A1 (en) | 2014-05-12 | 2015-11-19 | Virginia Tech Intellectual Properties, Inc. | Selective modulation of intracellular effects of cells using pulsed electric fields |
| US10045781B2 (en) | 2014-06-13 | 2018-08-14 | Ethicon Llc | Closure lockout systems for surgical instruments |
| CN105361944A (en) * | 2014-08-27 | 2016-03-02 | 上海微创电生理医疗科技有限公司 | Ablation catheter and method using same to implement ablation |
| US11311294B2 (en) | 2014-09-05 | 2022-04-26 | Cilag Gmbh International | Powered medical device including measurement of closure state of jaws |
| US10135242B2 (en) | 2014-09-05 | 2018-11-20 | Ethicon Llc | Smart cartridge wake up operation and data retention |
| BR112017004361B1 (en) | 2014-09-05 | 2023-04-11 | Ethicon Llc | ELECTRONIC SYSTEM FOR A SURGICAL INSTRUMENT |
| US10105142B2 (en) | 2014-09-18 | 2018-10-23 | Ethicon Llc | Surgical stapler with plurality of cutting elements |
| CN107427300B (en) | 2014-09-26 | 2020-12-04 | 伊西康有限责任公司 | Surgical suture buttress and buttress material |
| US11523821B2 (en) | 2014-09-26 | 2022-12-13 | Cilag Gmbh International | Method for creating a flexible staple line |
| US10076325B2 (en) | 2014-10-13 | 2018-09-18 | Ethicon Llc | Surgical stapling apparatus comprising a tissue stop |
| US9924944B2 (en) | 2014-10-16 | 2018-03-27 | Ethicon Llc | Staple cartridge comprising an adjunct material |
| US10517594B2 (en) | 2014-10-29 | 2019-12-31 | Ethicon Llc | Cartridge assemblies for surgical staplers |
| US11141153B2 (en) | 2014-10-29 | 2021-10-12 | Cilag Gmbh International | Staple cartridges comprising driver arrangements |
| US9844376B2 (en) | 2014-11-06 | 2017-12-19 | Ethicon Llc | Staple cartridge comprising a releasable adjunct material |
| WO2016080965A1 (en) * | 2014-11-18 | 2016-05-26 | Kardium Inc. | Systems and methods for activating transducers |
| US10736636B2 (en) | 2014-12-10 | 2020-08-11 | Ethicon Llc | Articulatable surgical instrument system |
| US10694972B2 (en) | 2014-12-15 | 2020-06-30 | Virginia Tech Intellectual Properties, Inc. | Devices, systems, and methods for real-time monitoring of electrophysical effects during tissue treatment |
| US9968355B2 (en) | 2014-12-18 | 2018-05-15 | Ethicon Llc | Surgical instruments with articulatable end effectors and improved firing beam support arrangements |
| US10085748B2 (en) | 2014-12-18 | 2018-10-02 | Ethicon Llc | Locking arrangements for detachable shaft assemblies with articulatable surgical end effectors |
| US10117649B2 (en) | 2014-12-18 | 2018-11-06 | Ethicon Llc | Surgical instrument assembly comprising a lockable articulation system |
| US9987000B2 (en) | 2014-12-18 | 2018-06-05 | Ethicon Llc | Surgical instrument assembly comprising a flexible articulation system |
| US9844375B2 (en) | 2014-12-18 | 2017-12-19 | Ethicon Llc | Drive arrangements for articulatable surgical instruments |
| BR112017012996B1 (en) | 2014-12-18 | 2022-11-08 | Ethicon Llc | SURGICAL INSTRUMENT WITH AN ANvil WHICH IS SELECTIVELY MOVABLE ABOUT AN IMMOVABLE GEOMETRIC AXIS DIFFERENT FROM A STAPLE CARTRIDGE |
| US10188385B2 (en) | 2014-12-18 | 2019-01-29 | Ethicon Llc | Surgical instrument system comprising lockable systems |
| US9844374B2 (en) | 2014-12-18 | 2017-12-19 | Ethicon Llc | Surgical instrument systems comprising an articulatable end effector and means for adjusting the firing stroke of a firing member |
| US10180463B2 (en) | 2015-02-27 | 2019-01-15 | Ethicon Llc | Surgical apparatus configured to assess whether a performance parameter of the surgical apparatus is within an acceptable performance band |
| US10226250B2 (en) | 2015-02-27 | 2019-03-12 | Ethicon Llc | Modular stapling assembly |
| US11154301B2 (en) | 2015-02-27 | 2021-10-26 | Cilag Gmbh International | Modular stapling assembly |
| US10321907B2 (en) | 2015-02-27 | 2019-06-18 | Ethicon Llc | System for monitoring whether a surgical instrument needs to be serviced |
| US9993248B2 (en) | 2015-03-06 | 2018-06-12 | Ethicon Endo-Surgery, Llc | Smart sensors with local signal processing |
| US9901342B2 (en) | 2015-03-06 | 2018-02-27 | Ethicon Endo-Surgery, Llc | Signal and power communication system positioned on a rotatable shaft |
| US10441279B2 (en) | 2015-03-06 | 2019-10-15 | Ethicon Llc | Multiple level thresholds to modify operation of powered surgical instruments |
| JP2020121162A (en) | 2015-03-06 | 2020-08-13 | エシコン エルエルシーEthicon LLC | Time dependent evaluation of sensor data to determine stability element, creep element and viscoelastic element of measurement |
| US9808246B2 (en) | 2015-03-06 | 2017-11-07 | Ethicon Endo-Surgery, Llc | Method of operating a powered surgical instrument |
| US10245033B2 (en) | 2015-03-06 | 2019-04-02 | Ethicon Llc | Surgical instrument comprising a lockable battery housing |
| US10687806B2 (en) | 2015-03-06 | 2020-06-23 | Ethicon Llc | Adaptive tissue compression techniques to adjust closure rates for multiple tissue types |
| US10045776B2 (en) | 2015-03-06 | 2018-08-14 | Ethicon Llc | Control techniques and sub-processor contained within modular shaft with select control processing from handle |
| US10617412B2 (en) | 2015-03-06 | 2020-04-14 | Ethicon Llc | System for detecting the mis-insertion of a staple cartridge into a surgical stapler |
| US10548504B2 (en) | 2015-03-06 | 2020-02-04 | Ethicon Llc | Overlaid multi sensor radio frequency (RF) electrode system to measure tissue compression |
| US9924961B2 (en) | 2015-03-06 | 2018-03-27 | Ethicon Endo-Surgery, Llc | Interactive feedback system for powered surgical instruments |
| US9895148B2 (en) | 2015-03-06 | 2018-02-20 | Ethicon Endo-Surgery, Llc | Monitoring speed control and precision incrementing of motor for powered surgical instruments |
| EP3274037B1 (en) | 2015-03-27 | 2021-11-03 | Kalila Medical, Inc. | Steerable medical devices |
| US10390825B2 (en) | 2015-03-31 | 2019-08-27 | Ethicon Llc | Surgical instrument with progressive rotary drive systems |
| WO2016172706A1 (en) | 2015-04-24 | 2016-10-27 | Shifamed Holdings, Llc | Steerable medical devices, systems, and methods of use |
| US10178992B2 (en) | 2015-06-18 | 2019-01-15 | Ethicon Llc | Push/pull articulation drive systems for articulatable surgical instruments |
| US10617418B2 (en) | 2015-08-17 | 2020-04-14 | Ethicon Llc | Implantable layers for a surgical instrument |
| US10357251B2 (en) | 2015-08-26 | 2019-07-23 | Ethicon Llc | Surgical staples comprising hardness variations for improved fastening of tissue |
| BR112018003693B1 (en) | 2015-08-26 | 2022-11-22 | Ethicon Llc | SURGICAL STAPLE CARTRIDGE FOR USE WITH A SURGICAL STAPPING INSTRUMENT |
| MX2022006192A (en) | 2015-09-02 | 2022-06-16 | Ethicon Llc | Surgical staple configurations with camming surfaces located between portions supporting surgical staples. |
| US10172619B2 (en) | 2015-09-02 | 2019-01-08 | Ethicon Llc | Surgical staple driver arrays |
| US10085751B2 (en) | 2015-09-23 | 2018-10-02 | Ethicon Llc | Surgical stapler having temperature-based motor control |
| US10105139B2 (en) | 2015-09-23 | 2018-10-23 | Ethicon Llc | Surgical stapler having downstream current-based motor control |
| US10363036B2 (en) | 2015-09-23 | 2019-07-30 | Ethicon Llc | Surgical stapler having force-based motor control |
| US10238386B2 (en) | 2015-09-23 | 2019-03-26 | Ethicon Llc | Surgical stapler having motor control based on an electrical parameter related to a motor current |
| US10076326B2 (en) | 2015-09-23 | 2018-09-18 | Ethicon Llc | Surgical stapler having current mirror-based motor control |
| US10327769B2 (en) | 2015-09-23 | 2019-06-25 | Ethicon Llc | Surgical stapler having motor control based on a drive system component |
| US10299878B2 (en) | 2015-09-25 | 2019-05-28 | Ethicon Llc | Implantable adjunct systems for determining adjunct skew |
| US11890015B2 (en) | 2015-09-30 | 2024-02-06 | Cilag Gmbh International | Compressible adjunct with crossing spacer fibers |
| US10980539B2 (en) | 2015-09-30 | 2021-04-20 | Ethicon Llc | Implantable adjunct comprising bonded layers |
| US10271849B2 (en) | 2015-09-30 | 2019-04-30 | Ethicon Llc | Woven constructs with interlocked standing fibers |
| US10524788B2 (en) | 2015-09-30 | 2020-01-07 | Ethicon Llc | Compressible adjunct with attachment regions |
| EP3373794B1 (en) | 2015-11-09 | 2022-01-05 | Kalila Medical, Inc. | Steering assemblies for medical devices |
| CN108348146A (en) | 2015-11-16 | 2018-07-31 | 阿帕玛医疗公司 | Energy transmission device |
| US10265068B2 (en) | 2015-12-30 | 2019-04-23 | Ethicon Llc | Surgical instruments with separable motors and motor control circuits |
| US10368865B2 (en) | 2015-12-30 | 2019-08-06 | Ethicon Llc | Mechanisms for compensating for drivetrain failure in powered surgical instruments |
| US10292704B2 (en) | 2015-12-30 | 2019-05-21 | Ethicon Llc | Mechanisms for compensating for battery pack failure in powered surgical instruments |
| CN108430368B (en) | 2016-01-07 | 2022-05-31 | 圣犹达医疗用品国际控股有限公司 | Medical device with multi-core optical fiber for optical sensing |
| US10588625B2 (en) | 2016-02-09 | 2020-03-17 | Ethicon Llc | Articulatable surgical instruments with off-axis firing beam arrangements |
| US11213293B2 (en) | 2016-02-09 | 2022-01-04 | Cilag Gmbh International | Articulatable surgical instruments with single articulation link arrangements |
| CN108882932B (en) | 2016-02-09 | 2021-07-23 | 伊西康有限责任公司 | Surgical instrument with asymmetric articulation configuration |
| US10258331B2 (en) | 2016-02-12 | 2019-04-16 | Ethicon Llc | Mechanisms for compensating for drivetrain failure in powered surgical instruments |
| US11224426B2 (en) | 2016-02-12 | 2022-01-18 | Cilag Gmbh International | Mechanisms for compensating for drivetrain failure in powered surgical instruments |
| US10448948B2 (en) | 2016-02-12 | 2019-10-22 | Ethicon Llc | Mechanisms for compensating for drivetrain failure in powered surgical instruments |
| US10617413B2 (en) | 2016-04-01 | 2020-04-14 | Ethicon Llc | Closure system arrangements for surgical cutting and stapling devices with separate and distinct firing shafts |
| US11064997B2 (en) | 2016-04-01 | 2021-07-20 | Cilag Gmbh International | Surgical stapling instrument |
| US10492783B2 (en) | 2016-04-15 | 2019-12-03 | Ethicon, Llc | Surgical instrument with improved stop/start control during a firing motion |
| US10335145B2 (en) | 2016-04-15 | 2019-07-02 | Ethicon Llc | Modular surgical instrument with configurable operating mode |
| US10357247B2 (en) | 2016-04-15 | 2019-07-23 | Ethicon Llc | Surgical instrument with multiple program responses during a firing motion |
| US10426467B2 (en) | 2016-04-15 | 2019-10-01 | Ethicon Llc | Surgical instrument with detection sensors |
| US10405859B2 (en) | 2016-04-15 | 2019-09-10 | Ethicon Llc | Surgical instrument with adjustable stop/start control during a firing motion |
| US10828028B2 (en) | 2016-04-15 | 2020-11-10 | Ethicon Llc | Surgical instrument with multiple program responses during a firing motion |
| US11607239B2 (en) | 2016-04-15 | 2023-03-21 | Cilag Gmbh International | Systems and methods for controlling a surgical stapling and cutting instrument |
| US10456137B2 (en) | 2016-04-15 | 2019-10-29 | Ethicon Llc | Staple formation detection mechanisms |
| US11179150B2 (en) | 2016-04-15 | 2021-11-23 | Cilag Gmbh International | Systems and methods for controlling a surgical stapling and cutting instrument |
| US10363037B2 (en) | 2016-04-18 | 2019-07-30 | Ethicon Llc | Surgical instrument system comprising a magnetic lockout |
| US20170296173A1 (en) | 2016-04-18 | 2017-10-19 | Ethicon Endo-Surgery, Llc | Method for operating a surgical instrument |
| US11317917B2 (en) | 2016-04-18 | 2022-05-03 | Cilag Gmbh International | Surgical stapling system comprising a lockable firing assembly |
| USD826405S1 (en) | 2016-06-24 | 2018-08-21 | Ethicon Llc | Surgical fastener |
| US10702270B2 (en) | 2016-06-24 | 2020-07-07 | Ethicon Llc | Stapling system for use with wire staples and stamped staples |
| USD850617S1 (en) | 2016-06-24 | 2019-06-04 | Ethicon Llc | Surgical fastener cartridge |
| USD847989S1 (en) | 2016-06-24 | 2019-05-07 | Ethicon Llc | Surgical fastener cartridge |
| CN109310431B (en) | 2016-06-24 | 2022-03-04 | 伊西康有限责任公司 | Staple cartridge comprising wire staples and punch staples |
| US10905492B2 (en) | 2016-11-17 | 2021-02-02 | Angiodynamics, Inc. | Techniques for irreversible electroporation using a single-pole tine-style internal device communicating with an external surface electrode |
| JP7010956B2 (en) | 2016-12-21 | 2022-01-26 | エシコン エルエルシー | How to staple tissue |
| US10537325B2 (en) | 2016-12-21 | 2020-01-21 | Ethicon Llc | Staple forming pocket arrangement to accommodate different types of staples |
| US10736629B2 (en) | 2016-12-21 | 2020-08-11 | Ethicon Llc | Surgical tool assemblies with clutching arrangements for shifting between closure systems with closure stroke reduction features and articulation and firing systems |
| US11419606B2 (en) | 2016-12-21 | 2022-08-23 | Cilag Gmbh International | Shaft assembly comprising a clutch configured to adapt the output of a rotary firing member to two different systems |
| US10492785B2 (en) | 2016-12-21 | 2019-12-03 | Ethicon Llc | Shaft assembly comprising a lockout |
| US20180168647A1 (en) | 2016-12-21 | 2018-06-21 | Ethicon Endo-Surgery, Llc | Surgical stapling instruments having end effectors with positive opening features |
| US10881401B2 (en) | 2016-12-21 | 2021-01-05 | Ethicon Llc | Staple firing member comprising a missing cartridge and/or spent cartridge lockout |
| US10568625B2 (en) | 2016-12-21 | 2020-02-25 | Ethicon Llc | Staple cartridges and arrangements of staples and staple cavities therein |
| US20180168615A1 (en) | 2016-12-21 | 2018-06-21 | Ethicon Endo-Surgery, Llc | Method of deforming staples from two different types of staple cartridges with the same surgical stapling instrument |
| US20180168598A1 (en) | 2016-12-21 | 2018-06-21 | Ethicon Endo-Surgery, Llc | Staple forming pocket arrangements comprising zoned forming surface grooves |
| US10485543B2 (en) | 2016-12-21 | 2019-11-26 | Ethicon Llc | Anvil having a knife slot width |
| US10993715B2 (en) | 2016-12-21 | 2021-05-04 | Ethicon Llc | Staple cartridge comprising staples with different clamping breadths |
| US11134942B2 (en) | 2016-12-21 | 2021-10-05 | Cilag Gmbh International | Surgical stapling instruments and staple-forming anvils |
| US10687810B2 (en) | 2016-12-21 | 2020-06-23 | Ethicon Llc | Stepped staple cartridge with tissue retention and gap setting features |
| US20180168609A1 (en) | 2016-12-21 | 2018-06-21 | Ethicon Endo-Surgery, Llc | Firing assembly comprising a fuse |
| US10973516B2 (en) | 2016-12-21 | 2021-04-13 | Ethicon Llc | Surgical end effectors and adaptable firing members therefor |
| JP6983893B2 (en) | 2016-12-21 | 2021-12-17 | エシコン エルエルシーEthicon LLC | Lockout configuration for surgical end effectors and replaceable tool assemblies |
| MX2019007311A (en) | 2016-12-21 | 2019-11-18 | Ethicon Llc | Surgical stapling systems. |
| US10945727B2 (en) | 2016-12-21 | 2021-03-16 | Ethicon Llc | Staple cartridge with deformable driver retention features |
| US11684367B2 (en) | 2016-12-21 | 2023-06-27 | Cilag Gmbh International | Stepped assembly having and end-of-life indicator |
| US10426471B2 (en) | 2016-12-21 | 2019-10-01 | Ethicon Llc | Surgical instrument with multiple failure response modes |
| US20180168575A1 (en) | 2016-12-21 | 2018-06-21 | Ethicon Endo-Surgery, Llc | Surgical stapling systems |
| US10980536B2 (en) | 2016-12-21 | 2021-04-20 | Ethicon Llc | No-cartridge and spent cartridge lockout arrangements for surgical staplers |
| US20180168625A1 (en) | 2016-12-21 | 2018-06-21 | Ethicon Endo-Surgery, Llc | Surgical stapling instruments with smart staple cartridges |
| EP3614942A2 (en) | 2017-04-28 | 2020-03-04 | Stryker Corporation | Control console and accessories for rf nerve ablation and methods of operating the same |
| USD879808S1 (en) | 2017-06-20 | 2020-03-31 | Ethicon Llc | Display panel with graphical user interface |
| US10881399B2 (en) | 2017-06-20 | 2021-01-05 | Ethicon Llc | Techniques for adaptive control of motor velocity of a surgical stapling and cutting instrument |
| US10881396B2 (en) | 2017-06-20 | 2021-01-05 | Ethicon Llc | Surgical instrument with variable duration trigger arrangement |
| US10368864B2 (en) | 2017-06-20 | 2019-08-06 | Ethicon Llc | Systems and methods for controlling displaying motor velocity for a surgical instrument |
| US10327767B2 (en) | 2017-06-20 | 2019-06-25 | Ethicon Llc | Control of motor velocity of a surgical stapling and cutting instrument based on angle of articulation |
| US10813639B2 (en) | 2017-06-20 | 2020-10-27 | Ethicon Llc | Closed loop feedback control of motor velocity of a surgical stapling and cutting instrument based on system conditions |
| US11090046B2 (en) | 2017-06-20 | 2021-08-17 | Cilag Gmbh International | Systems and methods for controlling displacement member motion of a surgical stapling and cutting instrument |
| US10624633B2 (en) | 2017-06-20 | 2020-04-21 | Ethicon Llc | Systems and methods for controlling motor velocity of a surgical stapling and cutting instrument |
| US11382638B2 (en) | 2017-06-20 | 2022-07-12 | Cilag Gmbh International | Closed loop feedback control of motor velocity of a surgical stapling and cutting instrument based on measured time over a specified displacement distance |
| USD890784S1 (en) | 2017-06-20 | 2020-07-21 | Ethicon Llc | Display panel with changeable graphical user interface |
| USD879809S1 (en) | 2017-06-20 | 2020-03-31 | Ethicon Llc | Display panel with changeable graphical user interface |
| US10779820B2 (en) | 2017-06-20 | 2020-09-22 | Ethicon Llc | Systems and methods for controlling motor speed according to user input for a surgical instrument |
| US10390841B2 (en) | 2017-06-20 | 2019-08-27 | Ethicon Llc | Control of motor velocity of a surgical stapling and cutting instrument based on angle of articulation |
| US11071554B2 (en) | 2017-06-20 | 2021-07-27 | Cilag Gmbh International | Closed loop feedback control of motor velocity of a surgical stapling and cutting instrument based on magnitude of velocity error measurements |
| US11517325B2 (en) | 2017-06-20 | 2022-12-06 | Cilag Gmbh International | Closed loop feedback control of motor velocity of a surgical stapling and cutting instrument based on measured displacement distance traveled over a specified time interval |
| US10980537B2 (en) | 2017-06-20 | 2021-04-20 | Ethicon Llc | Closed loop feedback control of motor velocity of a surgical stapling and cutting instrument based on measured time over a specified number of shaft rotations |
| US10307170B2 (en) | 2017-06-20 | 2019-06-04 | Ethicon Llc | Method for closed loop control of motor velocity of a surgical stapling and cutting instrument |
| US11653914B2 (en) | 2017-06-20 | 2023-05-23 | Cilag Gmbh International | Systems and methods for controlling motor velocity of a surgical stapling and cutting instrument according to articulation angle of end effector |
| US10646220B2 (en) | 2017-06-20 | 2020-05-12 | Ethicon Llc | Systems and methods for controlling displacement member velocity for a surgical instrument |
| US10888321B2 (en) | 2017-06-20 | 2021-01-12 | Ethicon Llc | Systems and methods for controlling velocity of a displacement member of a surgical stapling and cutting instrument |
| US11324503B2 (en) | 2017-06-27 | 2022-05-10 | Cilag Gmbh International | Surgical firing member arrangements |
| US10772629B2 (en) | 2017-06-27 | 2020-09-15 | Ethicon Llc | Surgical anvil arrangements |
| US10631859B2 (en) | 2017-06-27 | 2020-04-28 | Ethicon Llc | Articulation systems for surgical instruments |
| US11266405B2 (en) | 2017-06-27 | 2022-03-08 | Cilag Gmbh International | Surgical anvil manufacturing methods |
| US10856869B2 (en) | 2017-06-27 | 2020-12-08 | Ethicon Llc | Surgical anvil arrangements |
| US10993716B2 (en) | 2017-06-27 | 2021-05-04 | Ethicon Llc | Surgical anvil arrangements |
| US10765427B2 (en) | 2017-06-28 | 2020-09-08 | Ethicon Llc | Method for articulating a surgical instrument |
| USD851762S1 (en) | 2017-06-28 | 2019-06-18 | Ethicon Llc | Anvil |
| USD906355S1 (en) | 2017-06-28 | 2020-12-29 | Ethicon Llc | Display screen or portion thereof with a graphical user interface for a surgical instrument |
| US10211586B2 (en) | 2017-06-28 | 2019-02-19 | Ethicon Llc | Surgical shaft assemblies with watertight housings |
| USD869655S1 (en) | 2017-06-28 | 2019-12-10 | Ethicon Llc | Surgical fastener cartridge |
| US11246592B2 (en) | 2017-06-28 | 2022-02-15 | Cilag Gmbh International | Surgical instrument comprising an articulation system lockable to a frame |
| EP3420947B1 (en) | 2017-06-28 | 2022-05-25 | Cilag GmbH International | Surgical instrument comprising selectively actuatable rotatable couplers |
| US11564686B2 (en) | 2017-06-28 | 2023-01-31 | Cilag Gmbh International | Surgical shaft assemblies with flexible interfaces |
| US11389161B2 (en) | 2017-06-28 | 2022-07-19 | Cilag Gmbh International | Surgical instrument comprising selectively actuatable rotatable couplers |
| USD854151S1 (en) | 2017-06-28 | 2019-07-16 | Ethicon Llc | Surgical instrument shaft |
| US10716614B2 (en) | 2017-06-28 | 2020-07-21 | Ethicon Llc | Surgical shaft assemblies with slip ring assemblies with increased contact pressure |
| US10588633B2 (en) | 2017-06-28 | 2020-03-17 | Ethicon Llc | Surgical instruments with open and closable jaws and axially movable firing member that is initially parked in close proximity to the jaws prior to firing |
| US10903685B2 (en) | 2017-06-28 | 2021-01-26 | Ethicon Llc | Surgical shaft assemblies with slip ring assemblies forming capacitive channels |
| US11259805B2 (en) | 2017-06-28 | 2022-03-01 | Cilag Gmbh International | Surgical instrument comprising firing member supports |
| US10398434B2 (en) | 2017-06-29 | 2019-09-03 | Ethicon Llc | Closed loop velocity control of closure member for robotic surgical instrument |
| US10898183B2 (en) | 2017-06-29 | 2021-01-26 | Ethicon Llc | Robotic surgical instrument with closed loop feedback techniques for advancement of closure member during firing |
| US10932772B2 (en) | 2017-06-29 | 2021-03-02 | Ethicon Llc | Methods for closed loop velocity control for robotic surgical instrument |
| US10258418B2 (en) | 2017-06-29 | 2019-04-16 | Ethicon Llc | System for controlling articulation forces |
| US11007022B2 (en) | 2017-06-29 | 2021-05-18 | Ethicon Llc | Closed loop velocity control techniques based on sensed tissue parameters for robotic surgical instrument |
| US11666379B2 (en) | 2017-07-06 | 2023-06-06 | Biosense Webster (Israel) Ltd. | Temperature controlled short duration ablation with multiple electrodes |
| US11944300B2 (en) | 2017-08-03 | 2024-04-02 | Cilag Gmbh International | Method for operating a surgical system bailout |
| US11471155B2 (en) | 2017-08-03 | 2022-10-18 | Cilag Gmbh International | Surgical system bailout |
| US11304695B2 (en) | 2017-08-03 | 2022-04-19 | Cilag Gmbh International | Surgical system shaft interconnection |
| USD907648S1 (en) | 2017-09-29 | 2021-01-12 | Ethicon Llc | Display screen or portion thereof with animated graphical user interface |
| US11399829B2 (en) | 2017-09-29 | 2022-08-02 | Cilag Gmbh International | Systems and methods of initiating a power shutdown mode for a surgical instrument |
| US10796471B2 (en) | 2017-09-29 | 2020-10-06 | Ethicon Llc | Systems and methods of displaying a knife position for a surgical instrument |
| USD907647S1 (en) | 2017-09-29 | 2021-01-12 | Ethicon Llc | Display screen or portion thereof with animated graphical user interface |
| US10743872B2 (en) | 2017-09-29 | 2020-08-18 | Ethicon Llc | System and methods for controlling a display of a surgical instrument |
| USD917500S1 (en) | 2017-09-29 | 2021-04-27 | Ethicon Llc | Display screen or portion thereof with graphical user interface |
| US10729501B2 (en) | 2017-09-29 | 2020-08-04 | Ethicon Llc | Systems and methods for language selection of a surgical instrument |
| US10765429B2 (en) | 2017-09-29 | 2020-09-08 | Ethicon Llc | Systems and methods for providing alerts according to the operational state of a surgical instrument |
| US11090075B2 (en) | 2017-10-30 | 2021-08-17 | Cilag Gmbh International | Articulation features for surgical end effector |
| US11134944B2 (en) | 2017-10-30 | 2021-10-05 | Cilag Gmbh International | Surgical stapler knife motion controls |
| US10842490B2 (en) | 2017-10-31 | 2020-11-24 | Ethicon Llc | Cartridge body design with force reduction based on firing completion |
| US10779903B2 (en) | 2017-10-31 | 2020-09-22 | Ethicon Llc | Positive shaft rotation lock activated by jaw closure |
| US11607537B2 (en) | 2017-12-05 | 2023-03-21 | Virginia Tech Intellectual Properties, Inc. | Method for treating neurological disorders, including tumors, with electroporation |
| US11006955B2 (en) | 2017-12-15 | 2021-05-18 | Ethicon Llc | End effectors with positive jaw opening features for use with adapters for electromechanical surgical instruments |
| US10779825B2 (en) | 2017-12-15 | 2020-09-22 | Ethicon Llc | Adapters with end effector position sensing and control arrangements for use in connection with electromechanical surgical instruments |
| US10828033B2 (en) | 2017-12-15 | 2020-11-10 | Ethicon Llc | Handheld electromechanical surgical instruments with improved motor control arrangements for positioning components of an adapter coupled thereto |
| US10743875B2 (en) | 2017-12-15 | 2020-08-18 | Ethicon Llc | Surgical end effectors with jaw stiffener arrangements configured to permit monitoring of firing member |
| US10743874B2 (en) | 2017-12-15 | 2020-08-18 | Ethicon Llc | Sealed adapters for use with electromechanical surgical instruments |
| US10869666B2 (en) | 2017-12-15 | 2020-12-22 | Ethicon Llc | Adapters with control systems for controlling multiple motors of an electromechanical surgical instrument |
| US10966718B2 (en) | 2017-12-15 | 2021-04-06 | Ethicon Llc | Dynamic clamping assemblies with improved wear characteristics for use in connection with electromechanical surgical instruments |
| US10779826B2 (en) | 2017-12-15 | 2020-09-22 | Ethicon Llc | Methods of operating surgical end effectors |
| US10687813B2 (en) | 2017-12-15 | 2020-06-23 | Ethicon Llc | Adapters with firing stroke sensing arrangements for use in connection with electromechanical surgical instruments |
| US11197670B2 (en) | 2017-12-15 | 2021-12-14 | Cilag Gmbh International | Surgical end effectors with pivotal jaws configured to touch at their respective distal ends when fully closed |
| US11033267B2 (en) | 2017-12-15 | 2021-06-15 | Ethicon Llc | Systems and methods of controlling a clamping member firing rate of a surgical instrument |
| US11071543B2 (en) | 2017-12-15 | 2021-07-27 | Cilag Gmbh International | Surgical end effectors with clamping assemblies configured to increase jaw aperture ranges |
| US10729509B2 (en) | 2017-12-19 | 2020-08-04 | Ethicon Llc | Surgical instrument comprising closure and firing locking mechanism |
| USD910847S1 (en) | 2017-12-19 | 2021-02-16 | Ethicon Llc | Surgical instrument assembly |
| US11020112B2 (en) | 2017-12-19 | 2021-06-01 | Ethicon Llc | Surgical tools configured for interchangeable use with different controller interfaces |
| US10716565B2 (en) | 2017-12-19 | 2020-07-21 | Ethicon Llc | Surgical instruments with dual articulation drivers |
| US11045270B2 (en) | 2017-12-19 | 2021-06-29 | Cilag Gmbh International | Robotic attachment comprising exterior drive actuator |
| US10835330B2 (en) | 2017-12-19 | 2020-11-17 | Ethicon Llc | Method for determining the position of a rotatable jaw of a surgical instrument attachment assembly |
| US11311290B2 (en) | 2017-12-21 | 2022-04-26 | Cilag Gmbh International | Surgical instrument comprising an end effector dampener |
| US11129680B2 (en) | 2017-12-21 | 2021-09-28 | Cilag Gmbh International | Surgical instrument comprising a projector |
| US11751867B2 (en) | 2017-12-21 | 2023-09-12 | Cilag Gmbh International | Surgical instrument comprising sequenced systems |
| US11076853B2 (en) | 2017-12-21 | 2021-08-03 | Cilag Gmbh International | Systems and methods of displaying a knife position during transection for a surgical instrument |
| US11154352B2 (en) * | 2018-01-23 | 2021-10-26 | Biosense Webster (Israel) Ltd. | Power controlled short duration ablation with varying temperature limits |
| US11311329B2 (en) | 2018-03-13 | 2022-04-26 | Virginia Tech Intellectual Properties, Inc. | Treatment planning for immunotherapy based treatments using non-thermal ablation techniques |
| US11925405B2 (en) | 2018-03-13 | 2024-03-12 | Virginia Tech Intellectual Properties, Inc. | Treatment planning system for immunotherapy enhancement via non-thermal ablation |
| US11039834B2 (en) | 2018-08-20 | 2021-06-22 | Cilag Gmbh International | Surgical stapler anvils with staple directing protrusions and tissue stability features |
| US11045192B2 (en) | 2018-08-20 | 2021-06-29 | Cilag Gmbh International | Fabricating techniques for surgical stapler anvils |
| US10842492B2 (en) | 2018-08-20 | 2020-11-24 | Ethicon Llc | Powered articulatable surgical instruments with clutching and locking arrangements for linking an articulation drive system to a firing drive system |
| US10912559B2 (en) | 2018-08-20 | 2021-02-09 | Ethicon Llc | Reinforced deformable anvil tip for surgical stapler anvil |
| US10779821B2 (en) | 2018-08-20 | 2020-09-22 | Ethicon Llc | Surgical stapler anvils with tissue stop features configured to avoid tissue pinch |
| USD914878S1 (en) | 2018-08-20 | 2021-03-30 | Ethicon Llc | Surgical instrument anvil |
| US11083458B2 (en) | 2018-08-20 | 2021-08-10 | Cilag Gmbh International | Powered surgical instruments with clutching arrangements to convert linear drive motions to rotary drive motions |
| US11291440B2 (en) | 2018-08-20 | 2022-04-05 | Cilag Gmbh International | Method for operating a powered articulatable surgical instrument |
| US11207065B2 (en) | 2018-08-20 | 2021-12-28 | Cilag Gmbh International | Method for fabricating surgical stapler anvils |
| US11253256B2 (en) | 2018-08-20 | 2022-02-22 | Cilag Gmbh International | Articulatable motor powered surgical instruments with dedicated articulation motor arrangements |
| US10856870B2 (en) | 2018-08-20 | 2020-12-08 | Ethicon Llc | Switching arrangements for motor powered articulatable surgical instruments |
| US11324501B2 (en) | 2018-08-20 | 2022-05-10 | Cilag Gmbh International | Surgical stapling devices with improved closure members |
| US20210378735A1 (en) * | 2018-11-13 | 2021-12-09 | St. Jude Medical, Cardiology Division, Inc. | Phased array radiofrequency ablation catheter and method of its manufacture |
| EP3883642B1 (en) | 2018-11-19 | 2024-02-21 | Novocure GmbH | Arrays for delivering tumor treating fields (ttfields) with selectively addressable sub-elements |
| WO2020174429A1 (en) * | 2019-02-27 | 2020-09-03 | Yoram Wasserman | Delivering tumor treating fields (ttfields) using implantable transducer arrays |
| US11696761B2 (en) | 2019-03-25 | 2023-07-11 | Cilag Gmbh International | Firing drive arrangements for surgical systems |
| US11172929B2 (en) | 2019-03-25 | 2021-11-16 | Cilag Gmbh International | Articulation drive arrangements for surgical systems |
| US11147551B2 (en) | 2019-03-25 | 2021-10-19 | Cilag Gmbh International | Firing drive arrangements for surgical systems |
| US11147553B2 (en) | 2019-03-25 | 2021-10-19 | Cilag Gmbh International | Firing drive arrangements for surgical systems |
| US11471157B2 (en) | 2019-04-30 | 2022-10-18 | Cilag Gmbh International | Articulation control mapping for a surgical instrument |
| CN110063787B (en) * | 2019-04-30 | 2023-04-21 | 北京博海康源医疗器械有限公司 | Temperature control method and device of radio frequency ablation device |
| US11903581B2 (en) | 2019-04-30 | 2024-02-20 | Cilag Gmbh International | Methods for stapling tissue using a surgical instrument |
| US11452528B2 (en) | 2019-04-30 | 2022-09-27 | Cilag Gmbh International | Articulation actuators for a surgical instrument |
| US11432816B2 (en) | 2019-04-30 | 2022-09-06 | Cilag Gmbh International | Articulation pin for a surgical instrument |
| US11648009B2 (en) | 2019-04-30 | 2023-05-16 | Cilag Gmbh International | Rotatable jaw tip for a surgical instrument |
| US11253254B2 (en) | 2019-04-30 | 2022-02-22 | Cilag Gmbh International | Shaft rotation actuator on a surgical instrument |
| US11426251B2 (en) | 2019-04-30 | 2022-08-30 | Cilag Gmbh International | Articulation directional lights on a surgical instrument |
| US11684434B2 (en) | 2019-06-28 | 2023-06-27 | Cilag Gmbh International | Surgical RFID assemblies for instrument operational setting control |
| US11241235B2 (en) | 2019-06-28 | 2022-02-08 | Cilag Gmbh International | Method of using multiple RFID chips with a surgical assembly |
| US11638587B2 (en) | 2019-06-28 | 2023-05-02 | Cilag Gmbh International | RFID identification systems for surgical instruments |
| US11523822B2 (en) | 2019-06-28 | 2022-12-13 | Cilag Gmbh International | Battery pack including a circuit interrupter |
| US11259803B2 (en) | 2019-06-28 | 2022-03-01 | Cilag Gmbh International | Surgical stapling system having an information encryption protocol |
| US11224497B2 (en) | 2019-06-28 | 2022-01-18 | Cilag Gmbh International | Surgical systems with multiple RFID tags |
| US11627959B2 (en) | 2019-06-28 | 2023-04-18 | Cilag Gmbh International | Surgical instruments including manual and powered system lockouts |
| US11291451B2 (en) | 2019-06-28 | 2022-04-05 | Cilag Gmbh International | Surgical instrument with battery compatibility verification functionality |
| US11376098B2 (en) | 2019-06-28 | 2022-07-05 | Cilag Gmbh International | Surgical instrument system comprising an RFID system |
| US11298132B2 (en) | 2019-06-28 | 2022-04-12 | Cilag GmbH Inlernational | Staple cartridge including a honeycomb extension |
| US11246678B2 (en) | 2019-06-28 | 2022-02-15 | Cilag Gmbh International | Surgical stapling system having a frangible RFID tag |
| US11497492B2 (en) | 2019-06-28 | 2022-11-15 | Cilag Gmbh International | Surgical instrument including an articulation lock |
| US11660163B2 (en) | 2019-06-28 | 2023-05-30 | Cilag Gmbh International | Surgical system with RFID tags for updating motor assembly parameters |
| US11771419B2 (en) | 2019-06-28 | 2023-10-03 | Cilag Gmbh International | Packaging for a replaceable component of a surgical stapling system |
| US11298127B2 (en) | 2019-06-28 | 2022-04-12 | Cilag GmbH Interational | Surgical stapling system having a lockout mechanism for an incompatible cartridge |
| US11464601B2 (en) | 2019-06-28 | 2022-10-11 | Cilag Gmbh International | Surgical instrument comprising an RFID system for tracking a movable component |
| US11553971B2 (en) | 2019-06-28 | 2023-01-17 | Cilag Gmbh International | Surgical RFID assemblies for display and communication |
| US11426167B2 (en) | 2019-06-28 | 2022-08-30 | Cilag Gmbh International | Mechanisms for proper anvil attachment surgical stapling head assembly |
| US11219455B2 (en) | 2019-06-28 | 2022-01-11 | Cilag Gmbh International | Surgical instrument including a lockout key |
| US11478241B2 (en) | 2019-06-28 | 2022-10-25 | Cilag Gmbh International | Staple cartridge including projections |
| US11051807B2 (en) | 2019-06-28 | 2021-07-06 | Cilag Gmbh International | Packaging assembly including a particulate trap |
| US11399837B2 (en) | 2019-06-28 | 2022-08-02 | Cilag Gmbh International | Mechanisms for motor control adjustments of a motorized surgical instrument |
| US11931033B2 (en) | 2019-12-19 | 2024-03-19 | Cilag Gmbh International | Staple cartridge comprising a latch lockout |
| US11446029B2 (en) | 2019-12-19 | 2022-09-20 | Cilag Gmbh International | Staple cartridge comprising projections extending from a curved deck surface |
| US11529137B2 (en) | 2019-12-19 | 2022-12-20 | Cilag Gmbh International | Staple cartridge comprising driver retention members |
| US11529139B2 (en) | 2019-12-19 | 2022-12-20 | Cilag Gmbh International | Motor driven surgical instrument |
| US11607219B2 (en) | 2019-12-19 | 2023-03-21 | Cilag Gmbh International | Staple cartridge comprising a detachable tissue cutting knife |
| US11234698B2 (en) | 2019-12-19 | 2022-02-01 | Cilag Gmbh International | Stapling system comprising a clamp lockout and a firing lockout |
| US11464512B2 (en) | 2019-12-19 | 2022-10-11 | Cilag Gmbh International | Staple cartridge comprising a curved deck surface |
| US11701111B2 (en) | 2019-12-19 | 2023-07-18 | Cilag Gmbh International | Method for operating a surgical stapling instrument |
| US11291447B2 (en) | 2019-12-19 | 2022-04-05 | Cilag Gmbh International | Stapling instrument comprising independent jaw closing and staple firing systems |
| US11576672B2 (en) | 2019-12-19 | 2023-02-14 | Cilag Gmbh International | Surgical instrument comprising a closure system including a closure member and an opening member driven by a drive screw |
| US11304696B2 (en) | 2019-12-19 | 2022-04-19 | Cilag Gmbh International | Surgical instrument comprising a powered articulation system |
| US11844520B2 (en) | 2019-12-19 | 2023-12-19 | Cilag Gmbh International | Staple cartridge comprising driver retention members |
| US11911032B2 (en) | 2019-12-19 | 2024-02-27 | Cilag Gmbh International | Staple cartridge comprising a seating cam |
| US11559304B2 (en) | 2019-12-19 | 2023-01-24 | Cilag Gmbh International | Surgical instrument comprising a rapid closure mechanism |
| US11504122B2 (en) | 2019-12-19 | 2022-11-22 | Cilag Gmbh International | Surgical instrument comprising a nested firing member |
| USD967421S1 (en) | 2020-06-02 | 2022-10-18 | Cilag Gmbh International | Staple cartridge |
| USD975278S1 (en) | 2020-06-02 | 2023-01-10 | Cilag Gmbh International | Staple cartridge |
| USD976401S1 (en) | 2020-06-02 | 2023-01-24 | Cilag Gmbh International | Staple cartridge |
| US11839771B2 (en) | 2020-06-02 | 2023-12-12 | Novocure Gmbh | Implantable arrays for providing tumor treating fields |
| USD966512S1 (en) | 2020-06-02 | 2022-10-11 | Cilag Gmbh International | Staple cartridge |
| USD975850S1 (en) | 2020-06-02 | 2023-01-17 | Cilag Gmbh International | Staple cartridge |
| USD975851S1 (en) | 2020-06-02 | 2023-01-17 | Cilag Gmbh International | Staple cartridge |
| USD974560S1 (en) | 2020-06-02 | 2023-01-03 | Cilag Gmbh International | Staple cartridge |
| US20220031351A1 (en) | 2020-07-28 | 2022-02-03 | Cilag Gmbh International | Surgical instruments with differential articulation joint arrangements for accommodating flexible actuators |
| US11844518B2 (en) | 2020-10-29 | 2023-12-19 | Cilag Gmbh International | Method for operating a surgical instrument |
| US11517390B2 (en) | 2020-10-29 | 2022-12-06 | Cilag Gmbh International | Surgical instrument comprising a limited travel switch |
| US11534259B2 (en) | 2020-10-29 | 2022-12-27 | Cilag Gmbh International | Surgical instrument comprising an articulation indicator |
| US11617577B2 (en) | 2020-10-29 | 2023-04-04 | Cilag Gmbh International | Surgical instrument comprising a sensor configured to sense whether an articulation drive of the surgical instrument is actuatable |
| US11931025B2 (en) | 2020-10-29 | 2024-03-19 | Cilag Gmbh International | Surgical instrument comprising a releasable closure drive lock |
| USD980425S1 (en) | 2020-10-29 | 2023-03-07 | Cilag Gmbh International | Surgical instrument assembly |
| US11452526B2 (en) | 2020-10-29 | 2022-09-27 | Cilag Gmbh International | Surgical instrument comprising a staged voltage regulation start-up system |
| US11717289B2 (en) | 2020-10-29 | 2023-08-08 | Cilag Gmbh International | Surgical instrument comprising an indicator which indicates that an articulation drive is actuatable |
| US11779330B2 (en) | 2020-10-29 | 2023-10-10 | Cilag Gmbh International | Surgical instrument comprising a jaw alignment system |
| US11896217B2 (en) | 2020-10-29 | 2024-02-13 | Cilag Gmbh International | Surgical instrument comprising an articulation lock |
| USD1013170S1 (en) | 2020-10-29 | 2024-01-30 | Cilag Gmbh International | Surgical instrument assembly |
| US11849943B2 (en) | 2020-12-02 | 2023-12-26 | Cilag Gmbh International | Surgical instrument with cartridge release mechanisms |
| US11653920B2 (en) | 2020-12-02 | 2023-05-23 | Cilag Gmbh International | Powered surgical instruments with communication interfaces through sterile barrier |
| US11627960B2 (en) | 2020-12-02 | 2023-04-18 | Cilag Gmbh International | Powered surgical instruments with smart reload with separately attachable exteriorly mounted wiring connections |
| US11653915B2 (en) | 2020-12-02 | 2023-05-23 | Cilag Gmbh International | Surgical instruments with sled location detection and adjustment features |
| US11944296B2 (en) | 2020-12-02 | 2024-04-02 | Cilag Gmbh International | Powered surgical instruments with external connectors |
| US11737751B2 (en) | 2020-12-02 | 2023-08-29 | Cilag Gmbh International | Devices and methods of managing energy dissipated within sterile barriers of surgical instrument housings |
| US11744581B2 (en) | 2020-12-02 | 2023-09-05 | Cilag Gmbh International | Powered surgical instruments with multi-phase tissue treatment |
| US11890010B2 (en) | 2020-12-02 | 2024-02-06 | Cllag GmbH International | Dual-sided reinforced reload for surgical instruments |
| US11678882B2 (en) | 2020-12-02 | 2023-06-20 | Cilag Gmbh International | Surgical instruments with interactive features to remedy incidental sled movements |
| WO2022130183A2 (en) * | 2020-12-17 | 2022-06-23 | Novocure Gmbh | Pyroelectric-based temperature sensing of transducer arrays for applying tumor treating fields (ttfields) |
| US11793514B2 (en) | 2021-02-26 | 2023-10-24 | Cilag Gmbh International | Staple cartridge comprising sensor array which may be embedded in cartridge body |
| US11701113B2 (en) | 2021-02-26 | 2023-07-18 | Cilag Gmbh International | Stapling instrument comprising a separate power antenna and a data transfer antenna |
| US11696757B2 (en) | 2021-02-26 | 2023-07-11 | Cilag Gmbh International | Monitoring of internal systems to detect and track cartridge motion status |
| US11751869B2 (en) | 2021-02-26 | 2023-09-12 | Cilag Gmbh International | Monitoring of multiple sensors over time to detect moving characteristics of tissue |
| US11925349B2 (en) | 2021-02-26 | 2024-03-12 | Cilag Gmbh International | Adjustment to transfer parameters to improve available power |
| US11749877B2 (en) | 2021-02-26 | 2023-09-05 | Cilag Gmbh International | Stapling instrument comprising a signal antenna |
| US11723657B2 (en) | 2021-02-26 | 2023-08-15 | Cilag Gmbh International | Adjustable communication based on available bandwidth and power capacity |
| US11812964B2 (en) | 2021-02-26 | 2023-11-14 | Cilag Gmbh International | Staple cartridge comprising a power management circuit |
| US11730473B2 (en) | 2021-02-26 | 2023-08-22 | Cilag Gmbh International | Monitoring of manufacturing life-cycle |
| US11744583B2 (en) | 2021-02-26 | 2023-09-05 | Cilag Gmbh International | Distal communication array to tune frequency of RF systems |
| US11806011B2 (en) | 2021-03-22 | 2023-11-07 | Cilag Gmbh International | Stapling instrument comprising tissue compression systems |
| US11723658B2 (en) | 2021-03-22 | 2023-08-15 | Cilag Gmbh International | Staple cartridge comprising a firing lockout |
| US11826012B2 (en) | 2021-03-22 | 2023-11-28 | Cilag Gmbh International | Stapling instrument comprising a pulsed motor-driven firing rack |
| US11826042B2 (en) | 2021-03-22 | 2023-11-28 | Cilag Gmbh International | Surgical instrument comprising a firing drive including a selectable leverage mechanism |
| US11717291B2 (en) | 2021-03-22 | 2023-08-08 | Cilag Gmbh International | Staple cartridge comprising staples configured to apply different tissue compression |
| US11737749B2 (en) | 2021-03-22 | 2023-08-29 | Cilag Gmbh International | Surgical stapling instrument comprising a retraction system |
| US11759202B2 (en) | 2021-03-22 | 2023-09-19 | Cilag Gmbh International | Staple cartridge comprising an implantable layer |
| US11744603B2 (en) | 2021-03-24 | 2023-09-05 | Cilag Gmbh International | Multi-axis pivot joints for surgical instruments and methods for manufacturing same |
| US11786239B2 (en) | 2021-03-24 | 2023-10-17 | Cilag Gmbh International | Surgical instrument articulation joint arrangements comprising multiple moving linkage features |
| US11832816B2 (en) | 2021-03-24 | 2023-12-05 | Cilag Gmbh International | Surgical stapling assembly comprising nonplanar staples and planar staples |
| US11786243B2 (en) | 2021-03-24 | 2023-10-17 | Cilag Gmbh International | Firing members having flexible portions for adapting to a load during a surgical firing stroke |
| US11896219B2 (en) | 2021-03-24 | 2024-02-13 | Cilag Gmbh International | Mating features between drivers and underside of a cartridge deck |
| US11793516B2 (en) | 2021-03-24 | 2023-10-24 | Cilag Gmbh International | Surgical staple cartridge comprising longitudinal support beam |
| US11857183B2 (en) | 2021-03-24 | 2024-01-02 | Cilag Gmbh International | Stapling assembly components having metal substrates and plastic bodies |
| US11849945B2 (en) | 2021-03-24 | 2023-12-26 | Cilag Gmbh International | Rotary-driven surgical stapling assembly comprising eccentrically driven firing member |
| US11944336B2 (en) | 2021-03-24 | 2024-04-02 | Cilag Gmbh International | Joint arrangements for multi-planar alignment and support of operational drive shafts in articulatable surgical instruments |
| US11896218B2 (en) | 2021-03-24 | 2024-02-13 | Cilag Gmbh International | Method of using a powered stapling device |
| US11849944B2 (en) | 2021-03-24 | 2023-12-26 | Cilag Gmbh International | Drivers for fastener cartridge assemblies having rotary drive screws |
| US11903582B2 (en) | 2021-03-24 | 2024-02-20 | Cilag Gmbh International | Leveraging surfaces for cartridge installation |
| US11826047B2 (en) | 2021-05-28 | 2023-11-28 | Cilag Gmbh International | Stapling instrument comprising jaw mounts |
| US11877745B2 (en) | 2021-10-18 | 2024-01-23 | Cilag Gmbh International | Surgical stapling assembly having longitudinally-repeating staple leg clusters |
| US11937816B2 (en) | 2021-10-28 | 2024-03-26 | Cilag Gmbh International | Electrical lead arrangements for surgical instruments |
Citations (50)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4281373A (en) * | 1977-05-18 | 1981-07-28 | Societe Satelec | High frequency voltage generator |
| US4531524A (en) * | 1982-12-27 | 1985-07-30 | Rdm International, Inc. | Circuit apparatus and method for electrothermal treatment of cancer eye |
| US4559943A (en) * | 1981-09-03 | 1985-12-24 | C. R. Bard, Inc. | Electrosurgical generator |
| US4644955A (en) * | 1982-12-27 | 1987-02-24 | Rdm International, Inc. | Circuit apparatus and method for electrothermal treatment of cancer eye |
| US4739759A (en) * | 1985-02-26 | 1988-04-26 | Concept, Inc. | Microprocessor controlled electrosurgical generator |
| US4907589A (en) * | 1988-04-29 | 1990-03-13 | Cosman Eric R | Automatic over-temperature control apparatus for a therapeutic heating device |
| US4936281A (en) * | 1989-04-13 | 1990-06-26 | Everest Medical Corporation | Ultrasonically enhanced RF ablation catheter |
| US5122137A (en) * | 1990-04-27 | 1992-06-16 | Boston Scientific Corporation | Temperature controlled rf coagulation |
| US5233515A (en) * | 1990-06-08 | 1993-08-03 | Cosman Eric R | Real-time graphic display of heat lesioning parameters in a clinical lesion generator system |
| US5300068A (en) * | 1992-04-21 | 1994-04-05 | St. Jude Medical, Inc. | Electrosurgical apparatus |
| US5372596A (en) * | 1993-07-27 | 1994-12-13 | Valleylab Inc. | Apparatus for leakage control and method for its use |
| US5383917A (en) * | 1991-07-05 | 1995-01-24 | Jawahar M. Desai | Device and method for multi-phase radio-frequency ablation |
| US5423808A (en) * | 1991-11-08 | 1995-06-13 | Ep Technologies, Inc. | Systems and methods for radiofrequency ablation with phase sensitive power detection |
| US5433740A (en) * | 1991-04-25 | 1995-07-18 | Olympus Optical Co., Ltd. | Method and apparatus for thermotherapy |
| US5438302A (en) * | 1993-07-12 | 1995-08-01 | Gyrus Medical Limited | Electrosurgical radiofrequency generator having regulated voltage across switching device |
| US5437664A (en) * | 1994-01-18 | 1995-08-01 | Endovascular, Inc. | Apparatus and method for venous ligation |
| US5476495A (en) * | 1993-03-16 | 1995-12-19 | Ep Technologies, Inc. | Cardiac mapping and ablation systems |
| US5487385A (en) * | 1993-12-03 | 1996-01-30 | Avitall; Boaz | Atrial mapping and ablation catheter system |
| US5496312A (en) * | 1993-10-07 | 1996-03-05 | Valleylab Inc. | Impedance and temperature generator control |
| US5500011A (en) * | 1986-11-14 | 1996-03-19 | Desai; Jawahar M. | Catheter for mapping and ablation and method therefor |
| US5514130A (en) * | 1994-10-11 | 1996-05-07 | Dorsal Med International | RF apparatus for controlled depth ablation of soft tissue |
| US5540681A (en) * | 1992-04-10 | 1996-07-30 | Medtronic Cardiorhythm | Method and system for radiofrequency ablation of tissue |
| US5540684A (en) * | 1994-07-28 | 1996-07-30 | Hassler, Jr.; William L. | Method and apparatus for electrosurgically treating tissue |
| US5542916A (en) * | 1992-08-12 | 1996-08-06 | Vidamed, Inc. | Dual-channel RF power delivery system |
| US5562720A (en) * | 1992-05-01 | 1996-10-08 | Vesta Medical, Inc. | Bipolar/monopolar endometrial ablation device and method |
| US5573533A (en) * | 1992-04-10 | 1996-11-12 | Medtronic Cardiorhythm | Method and system for radiofrequency ablation of cardiac tissue |
| US5582609A (en) * | 1993-10-14 | 1996-12-10 | Ep Technologies, Inc. | Systems and methods for forming large lesions in body tissue using curvilinear electrode elements |
| US5584830A (en) * | 1994-03-30 | 1996-12-17 | Medtronic Cardiorhythm | Method and system for radiofrequency ablation of cardiac tissue |
| US5617854A (en) * | 1994-06-22 | 1997-04-08 | Munsif; Anand | Shaped catheter device and method |
| US5620481A (en) * | 1991-07-05 | 1997-04-15 | Desai; Jawahar M. | Device for multi-phase radio-frequency ablation |
| US5643197A (en) * | 1993-12-21 | 1997-07-01 | Angeion Corporation | Fluid cooled and perfused tip for a catheter |
| US5697928A (en) * | 1996-09-23 | 1997-12-16 | Uab Research Foundation | Cardic electrode catheter |
| US5702386A (en) * | 1991-11-08 | 1997-12-30 | Ep Technologies, Inc. | Non-linear control systems and methods for heating and ablating body tissue |
| US5769847A (en) * | 1994-06-27 | 1998-06-23 | Ep Technologies, Inc. | Systems and methods for controlling tissue ablation using multiple temperature sensing elements |
| US5797903A (en) * | 1996-04-12 | 1998-08-25 | Ep Technologies, Inc. | Tissue heating and ablation systems and methods using porous electrode structures with electrically conductive surfaces |
| US5810802A (en) * | 1994-08-08 | 1998-09-22 | E.P. Technologies, Inc. | Systems and methods for controlling tissue ablation using multiple temperature sensing elements |
| US5827273A (en) * | 1994-06-24 | 1998-10-27 | Stuart D. Edwards | Thin layer ablation apparatus |
| US5837001A (en) * | 1995-12-08 | 1998-11-17 | C. R. Bard | Radio frequency energy delivery system for multipolar electrode catheters |
| US5954719A (en) * | 1996-12-11 | 1999-09-21 | Irvine Biomedical, Inc. | System for operating a RF ablation generator |
| US6042580A (en) * | 1998-05-05 | 2000-03-28 | Cardiac Pacemakers, Inc. | Electrode having composition-matched, common-lead thermocouple wire for providing multiple temperature-sensitive junctions |
| US6045550A (en) * | 1998-05-05 | 2000-04-04 | Cardiac Peacemakers, Inc. | Electrode having non-joined thermocouple for providing multiple temperature-sensitive junctions |
| US6050994A (en) * | 1998-05-05 | 2000-04-18 | Cardiac Pacemakers, Inc. | RF ablation apparatus and method using controllable duty cycle with alternate phasing |
| US6059778A (en) * | 1998-05-05 | 2000-05-09 | Cardiac Pacemakers, Inc. | RF ablation apparatus and method using unipolar and bipolar techniques |
| US6123702A (en) * | 1998-09-10 | 2000-09-26 | Scimed Life Systems, Inc. | Systems and methods for controlling power in an electrosurgical probe |
| US6132426A (en) * | 1998-05-05 | 2000-10-17 | Daig Corporation | Temperature and current limited ablation catheter |
| US6139546A (en) * | 1997-10-06 | 2000-10-31 | Somnus Medical Technologies, Inc. | Linear power control with digital phase lock |
| US6162217A (en) * | 1999-04-21 | 2000-12-19 | Oratec Interventions, Inc. | Method and apparatus for controlling a temperature-controlled probe |
| US6171305B1 (en) * | 1998-05-05 | 2001-01-09 | Cardiac Pacemakers, Inc. | RF ablation apparatus and method having high output impedance drivers |
| US6183468B1 (en) * | 1998-09-10 | 2001-02-06 | Scimed Life Systems, Inc. | Systems and methods for controlling power in an electrosurgical probe |
| US6468270B1 (en) * | 1995-06-07 | 2002-10-22 | Arthocare Corporation | System and method for electrosurgical treatment of intervertebral discs |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6049737A (en) | 1998-05-05 | 2000-04-11 | Cardiac Pacemakers, Inc. | Catheter having common lead for electrode and sensor |
-
2000
- 2000-12-13 US US09/738,032 patent/US6558378B2/en not_active Expired - Lifetime
-
2001
- 2001-12-11 WO PCT/US2001/048220 patent/WO2002047565A2/en not_active Application Discontinuation
- 2001-12-11 AU AU2002230812A patent/AU2002230812A1/en not_active Abandoned
-
2003
- 2003-03-27 US US10/400,770 patent/US20030195501A1/en not_active Abandoned
Patent Citations (52)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4281373A (en) * | 1977-05-18 | 1981-07-28 | Societe Satelec | High frequency voltage generator |
| US4559943A (en) * | 1981-09-03 | 1985-12-24 | C. R. Bard, Inc. | Electrosurgical generator |
| US4644955A (en) * | 1982-12-27 | 1987-02-24 | Rdm International, Inc. | Circuit apparatus and method for electrothermal treatment of cancer eye |
| US4531524A (en) * | 1982-12-27 | 1985-07-30 | Rdm International, Inc. | Circuit apparatus and method for electrothermal treatment of cancer eye |
| US4739759A (en) * | 1985-02-26 | 1988-04-26 | Concept, Inc. | Microprocessor controlled electrosurgical generator |
| US5500011A (en) * | 1986-11-14 | 1996-03-19 | Desai; Jawahar M. | Catheter for mapping and ablation and method therefor |
| US4907589A (en) * | 1988-04-29 | 1990-03-13 | Cosman Eric R | Automatic over-temperature control apparatus for a therapeutic heating device |
| US4936281A (en) * | 1989-04-13 | 1990-06-26 | Everest Medical Corporation | Ultrasonically enhanced RF ablation catheter |
| US5122137A (en) * | 1990-04-27 | 1992-06-16 | Boston Scientific Corporation | Temperature controlled rf coagulation |
| US5233515A (en) * | 1990-06-08 | 1993-08-03 | Cosman Eric R | Real-time graphic display of heat lesioning parameters in a clinical lesion generator system |
| US5433740A (en) * | 1991-04-25 | 1995-07-18 | Olympus Optical Co., Ltd. | Method and apparatus for thermotherapy |
| US5383917A (en) * | 1991-07-05 | 1995-01-24 | Jawahar M. Desai | Device and method for multi-phase radio-frequency ablation |
| US5620481A (en) * | 1991-07-05 | 1997-04-15 | Desai; Jawahar M. | Device for multi-phase radio-frequency ablation |
| US5423808A (en) * | 1991-11-08 | 1995-06-13 | Ep Technologies, Inc. | Systems and methods for radiofrequency ablation with phase sensitive power detection |
| US5702386A (en) * | 1991-11-08 | 1997-12-30 | Ep Technologies, Inc. | Non-linear control systems and methods for heating and ablating body tissue |
| US5540681A (en) * | 1992-04-10 | 1996-07-30 | Medtronic Cardiorhythm | Method and system for radiofrequency ablation of tissue |
| US5573533A (en) * | 1992-04-10 | 1996-11-12 | Medtronic Cardiorhythm | Method and system for radiofrequency ablation of cardiac tissue |
| US5300068A (en) * | 1992-04-21 | 1994-04-05 | St. Jude Medical, Inc. | Electrosurgical apparatus |
| US5562720A (en) * | 1992-05-01 | 1996-10-08 | Vesta Medical, Inc. | Bipolar/monopolar endometrial ablation device and method |
| US5542916A (en) * | 1992-08-12 | 1996-08-06 | Vidamed, Inc. | Dual-channel RF power delivery system |
| US5476495A (en) * | 1993-03-16 | 1995-12-19 | Ep Technologies, Inc. | Cardiac mapping and ablation systems |
| US5438302A (en) * | 1993-07-12 | 1995-08-01 | Gyrus Medical Limited | Electrosurgical radiofrequency generator having regulated voltage across switching device |
| US5372596A (en) * | 1993-07-27 | 1994-12-13 | Valleylab Inc. | Apparatus for leakage control and method for its use |
| US5496312A (en) * | 1993-10-07 | 1996-03-05 | Valleylab Inc. | Impedance and temperature generator control |
| US5582609A (en) * | 1993-10-14 | 1996-12-10 | Ep Technologies, Inc. | Systems and methods for forming large lesions in body tissue using curvilinear electrode elements |
| US5487385A (en) * | 1993-12-03 | 1996-01-30 | Avitall; Boaz | Atrial mapping and ablation catheter system |
| US5643197A (en) * | 1993-12-21 | 1997-07-01 | Angeion Corporation | Fluid cooled and perfused tip for a catheter |
| US5437664A (en) * | 1994-01-18 | 1995-08-01 | Endovascular, Inc. | Apparatus and method for venous ligation |
| US5584830A (en) * | 1994-03-30 | 1996-12-17 | Medtronic Cardiorhythm | Method and system for radiofrequency ablation of cardiac tissue |
| US5617854A (en) * | 1994-06-22 | 1997-04-08 | Munsif; Anand | Shaped catheter device and method |
| US5827273A (en) * | 1994-06-24 | 1998-10-27 | Stuart D. Edwards | Thin layer ablation apparatus |
| US5769847A (en) * | 1994-06-27 | 1998-06-23 | Ep Technologies, Inc. | Systems and methods for controlling tissue ablation using multiple temperature sensing elements |
| US5540684A (en) * | 1994-07-28 | 1996-07-30 | Hassler, Jr.; William L. | Method and apparatus for electrosurgically treating tissue |
| US5810802A (en) * | 1994-08-08 | 1998-09-22 | E.P. Technologies, Inc. | Systems and methods for controlling tissue ablation using multiple temperature sensing elements |
| US5514130A (en) * | 1994-10-11 | 1996-05-07 | Dorsal Med International | RF apparatus for controlled depth ablation of soft tissue |
| US6468270B1 (en) * | 1995-06-07 | 2002-10-22 | Arthocare Corporation | System and method for electrosurgical treatment of intervertebral discs |
| US5837001A (en) * | 1995-12-08 | 1998-11-17 | C. R. Bard | Radio frequency energy delivery system for multipolar electrode catheters |
| US5797903A (en) * | 1996-04-12 | 1998-08-25 | Ep Technologies, Inc. | Tissue heating and ablation systems and methods using porous electrode structures with electrically conductive surfaces |
| US5697928A (en) * | 1996-09-23 | 1997-12-16 | Uab Research Foundation | Cardic electrode catheter |
| US5954719A (en) * | 1996-12-11 | 1999-09-21 | Irvine Biomedical, Inc. | System for operating a RF ablation generator |
| US6139546A (en) * | 1997-10-06 | 2000-10-31 | Somnus Medical Technologies, Inc. | Linear power control with digital phase lock |
| US6059778A (en) * | 1998-05-05 | 2000-05-09 | Cardiac Pacemakers, Inc. | RF ablation apparatus and method using unipolar and bipolar techniques |
| US6042580A (en) * | 1998-05-05 | 2000-03-28 | Cardiac Pacemakers, Inc. | Electrode having composition-matched, common-lead thermocouple wire for providing multiple temperature-sensitive junctions |
| US6132426A (en) * | 1998-05-05 | 2000-10-17 | Daig Corporation | Temperature and current limited ablation catheter |
| US6050994A (en) * | 1998-05-05 | 2000-04-18 | Cardiac Pacemakers, Inc. | RF ablation apparatus and method using controllable duty cycle with alternate phasing |
| US6171305B1 (en) * | 1998-05-05 | 2001-01-09 | Cardiac Pacemakers, Inc. | RF ablation apparatus and method having high output impedance drivers |
| US6200314B1 (en) * | 1998-05-05 | 2001-03-13 | Cardiac Pacemakers, Inc. | RF ablation apparatus and method using unipolar and bipolar techniques |
| US6309385B1 (en) * | 1998-05-05 | 2001-10-30 | Cardiac Pacemakers, Inc. | Electrode having composition-matched, common-lead thermocouple wire for providing multiple temperature-sensitive junctions |
| US6045550A (en) * | 1998-05-05 | 2000-04-04 | Cardiac Peacemakers, Inc. | Electrode having non-joined thermocouple for providing multiple temperature-sensitive junctions |
| US6123702A (en) * | 1998-09-10 | 2000-09-26 | Scimed Life Systems, Inc. | Systems and methods for controlling power in an electrosurgical probe |
| US6183468B1 (en) * | 1998-09-10 | 2001-02-06 | Scimed Life Systems, Inc. | Systems and methods for controlling power in an electrosurgical probe |
| US6162217A (en) * | 1999-04-21 | 2000-12-19 | Oratec Interventions, Inc. | Method and apparatus for controlling a temperature-controlled probe |
Cited By (147)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8361068B2 (en) | 2000-03-06 | 2013-01-29 | Medtronic Advanced Energy Llc | Fluid-assisted electrosurgical devices, electrosurgical unit with pump and methods of use thereof |
| US7922714B2 (en) | 2003-03-28 | 2011-04-12 | C.R. Bard, Inc. | Method and apparatus for selecting operating parameter values in electrophysiology procedures |
| US20070167940A1 (en) * | 2003-03-28 | 2007-07-19 | Debbie Stevens-Wright | Method and apparatus for selecting operating parameter values in electrophysiology procedures |
| US9125666B2 (en) | 2003-09-12 | 2015-09-08 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation of atherosclerotic material |
| US9510901B2 (en) | 2003-09-12 | 2016-12-06 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation |
| US10188457B2 (en) | 2003-09-12 | 2019-01-29 | Vessix Vascular, Inc. | Selectable eccentric remodeling and/or ablation |
| US8939970B2 (en) | 2004-09-10 | 2015-01-27 | Vessix Vascular, Inc. | Tuned RF energy and electrical tissue characterization for selective treatment of target tissues |
| US9125667B2 (en) | 2004-09-10 | 2015-09-08 | Vessix Vascular, Inc. | System for inducing desirable temperature effects on body tissue |
| US9713730B2 (en) | 2004-09-10 | 2017-07-25 | Boston Scientific Scimed, Inc. | Apparatus and method for treatment of in-stent restenosis |
| US8920414B2 (en) | 2004-09-10 | 2014-12-30 | Vessix Vascular, Inc. | Tuned RF energy and electrical tissue characterization for selective treatment of target tissues |
| US9642675B2 (en) | 2004-10-14 | 2017-05-09 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US8486063B2 (en) | 2004-10-14 | 2013-07-16 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US8617152B2 (en) | 2004-11-15 | 2013-12-31 | Medtronic Ablation Frontiers Llc | Ablation system with feedback |
| US9005194B2 (en) | 2004-11-24 | 2015-04-14 | Medtronic Ablation Frontiers Llc | Atrial ablation catheter adapted for treatment of septal wall arrhythmogenic foci and method of use |
| US8273084B2 (en) | 2004-11-24 | 2012-09-25 | Medtronic Ablation Frontiers Llc | Atrial ablation catheter and method of use |
| US8364237B2 (en) | 2005-03-28 | 2013-01-29 | Vessix Vascular, Inc. | Tuned RF energy for selective treatment of atheroma and other target tissues and/or structures |
| US7742795B2 (en) * | 2005-03-28 | 2010-06-22 | Minnow Medical, Inc. | Tuned RF energy for selective treatment of atheroma and other target tissues and/or structures |
| US20060223455A1 (en) * | 2005-03-31 | 2006-10-05 | Silicon Laboratories Inc. | System and method for wireless communication using low-pulling digital interface signals |
| US9486355B2 (en) | 2005-05-03 | 2016-11-08 | Vessix Vascular, Inc. | Selective accumulation of energy with or without knowledge of tissue topography |
| US8771267B2 (en) | 2005-06-20 | 2014-07-08 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US8337492B2 (en) | 2005-06-20 | 2012-12-25 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US7850685B2 (en) | 2005-06-20 | 2010-12-14 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US8979841B2 (en) | 2005-06-20 | 2015-03-17 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US9468495B2 (en) | 2005-06-20 | 2016-10-18 | Medtronic Ablation Frontiers Llc | Ablation catheter |
| US9566113B2 (en) | 2005-07-11 | 2017-02-14 | Medtronic Ablation Frontiers Llc | Low power tissue ablation system |
| US8834461B2 (en) | 2005-07-11 | 2014-09-16 | Medtronic Ablation Frontiers Llc | Low power tissue ablation system |
| US8657814B2 (en) | 2005-08-22 | 2014-02-25 | Medtronic Ablation Frontiers Llc | User interface for tissue ablation system |
| US9808300B2 (en) | 2006-05-02 | 2017-11-07 | Boston Scientific Scimed, Inc. | Control of arterial smooth muscle tone |
| US8034052B2 (en) | 2006-05-05 | 2011-10-11 | Covidien Ag | Apparatus and method for electrode thermosurgery |
| US7846158B2 (en) * | 2006-05-05 | 2010-12-07 | Covidien Ag | Apparatus and method for electrode thermosurgery |
| US10413356B2 (en) | 2006-10-18 | 2019-09-17 | Boston Scientific Scimed, Inc. | System for inducing desirable temperature effects on body tissue |
| US10213252B2 (en) | 2006-10-18 | 2019-02-26 | Vessix, Inc. | Inducing desirable temperature effects on body tissue |
| US9974607B2 (en) | 2006-10-18 | 2018-05-22 | Vessix Vascular, Inc. | Inducing desirable temperature effects on body tissue |
| EP2088951A4 (en) * | 2006-12-07 | 2010-05-19 | Terumo Corp | Apparatus and methods for multipolar tissue welding |
| US20080300590A1 (en) * | 2006-12-07 | 2008-12-04 | Cierra, Inc. | Apparatus and methods for multipolar tissue welding |
| EP2088951A1 (en) * | 2006-12-07 | 2009-08-19 | Terumo Kabushiki Kaisha | Apparatus and methods for multipolar tissue welding |
| US8496653B2 (en) | 2007-04-23 | 2013-07-30 | Boston Scientific Scimed, Inc. | Thrombus removal |
| US8771269B2 (en) | 2007-05-11 | 2014-07-08 | Medtronic Ablation Frontiers Llc | RF energy delivery system and method |
| US8641704B2 (en) | 2007-05-11 | 2014-02-04 | Medtronic Ablation Frontiers Llc | Ablation therapy system and method for treating continuous atrial fibrillation |
| US10219857B2 (en) | 2007-05-11 | 2019-03-05 | Medtronic Ablation Frontiers Llc | RF energy delivery system |
| US20100030210A1 (en) * | 2008-08-01 | 2010-02-04 | Paulus Joseph A | Polyphase Electrosurgical System and Method |
| US9700366B2 (en) * | 2008-08-01 | 2017-07-11 | Covidien Lp | Polyphase electrosurgical system and method |
| WO2010051305A1 (en) * | 2008-10-28 | 2010-05-06 | Smith & Nephew, Inc. | Electrosurgical device with controllable electric field profile |
| US20100145329A1 (en) * | 2008-10-28 | 2010-06-10 | Smith & Nephew, Inc. | Electrosurgical Device with Controllable Electric Field Profile |
| US8388615B2 (en) | 2008-10-28 | 2013-03-05 | Smith & Nephew, Inc. | Electrosurgical device with controllable electric field profile |
| CN102202591A (en) * | 2008-10-28 | 2011-09-28 | 史密夫和内修有限公司 | Electrosurgical device with controllable electric field profile |
| US8500729B2 (en) | 2008-10-28 | 2013-08-06 | Smith & Nephew, Inc. | Electrosurgical device with controllable electric field profile |
| US8295902B2 (en) | 2008-11-11 | 2012-10-23 | Shifamed Holdings, Llc | Low profile electrode assembly |
| US8396548B2 (en) | 2008-11-14 | 2013-03-12 | Vessix Vascular, Inc. | Selective drug delivery in a lumen |
| US9327100B2 (en) | 2008-11-14 | 2016-05-03 | Vessix Vascular, Inc. | Selective drug delivery in a lumen |
| US8401667B2 (en) | 2008-11-17 | 2013-03-19 | Vessix Vascular, Inc. | Selective accumulation of energy with or without knowledge of tissue topography |
| US8551096B2 (en) | 2009-05-13 | 2013-10-08 | Boston Scientific Scimed, Inc. | Directional delivery of energy and bioactives |
| US10327845B2 (en) | 2010-01-25 | 2019-06-25 | Covidien Lp | System and method for monitoring ablation size |
| US20140296841A1 (en) * | 2010-01-25 | 2014-10-02 | Covidien Lp | System and method for monitoring ablation size |
| US9820813B2 (en) * | 2010-01-25 | 2017-11-21 | Covidien Lp | System and method for monitoring ablation size |
| US20110218526A1 (en) * | 2010-03-03 | 2011-09-08 | Medtronic Ablation Frontiers, Llc | Variable-output radiofrequency ablation power supply |
| US8556891B2 (en) | 2010-03-03 | 2013-10-15 | Medtronic Ablation Frontiers Llc | Variable-output radiofrequency ablation power supply |
| CN102811677A (en) * | 2010-03-03 | 2012-12-05 | 麦德托尼克消融前沿有限公司 | Variable-output radiofrequency ablation power supply |
| US9155590B2 (en) | 2010-03-03 | 2015-10-13 | Medtronic Ablation Frontiers, Llc | Variable-output radiofrequency ablation power supply |
| WO2011109141A1 (en) * | 2010-03-03 | 2011-09-09 | Medtronic Ablation Frontiers Llc | Variable-output radiofrequency ablation power supply |
| US9277955B2 (en) | 2010-04-09 | 2016-03-08 | Vessix Vascular, Inc. | Power generating and control apparatus for the treatment of tissue |
| US9192790B2 (en) | 2010-04-14 | 2015-11-24 | Boston Scientific Scimed, Inc. | Focused ultrasonic renal denervation |
| US8880185B2 (en) | 2010-06-11 | 2014-11-04 | Boston Scientific Scimed, Inc. | Renal denervation and stimulation employing wireless vascular energy transfer arrangement |
| US9155589B2 (en) | 2010-07-30 | 2015-10-13 | Boston Scientific Scimed, Inc. | Sequential activation RF electrode set for renal nerve ablation |
| US9084609B2 (en) | 2010-07-30 | 2015-07-21 | Boston Scientific Scime, Inc. | Spiral balloon catheter for renal nerve ablation |
| US9463062B2 (en) | 2010-07-30 | 2016-10-11 | Boston Scientific Scimed, Inc. | Cooled conductive balloon RF catheter for renal nerve ablation |
| US9408661B2 (en) | 2010-07-30 | 2016-08-09 | Patrick A. Haverkost | RF electrodes on multiple flexible wires for renal nerve ablation |
| US9358365B2 (en) | 2010-07-30 | 2016-06-07 | Boston Scientific Scimed, Inc. | Precision electrode movement control for renal nerve ablation |
| US8974451B2 (en) | 2010-10-25 | 2015-03-10 | Boston Scientific Scimed, Inc. | Renal nerve ablation using conductive fluid jet and RF energy |
| US9220558B2 (en) | 2010-10-27 | 2015-12-29 | Boston Scientific Scimed, Inc. | RF renal denervation catheter with multiple independent electrodes |
| US9848946B2 (en) | 2010-11-15 | 2017-12-26 | Boston Scientific Scimed, Inc. | Self-expanding cooling electrode for renal nerve ablation |
| US9028485B2 (en) | 2010-11-15 | 2015-05-12 | Boston Scientific Scimed, Inc. | Self-expanding cooling electrode for renal nerve ablation |
| US9089350B2 (en) | 2010-11-16 | 2015-07-28 | Boston Scientific Scimed, Inc. | Renal denervation catheter with RF electrode and integral contrast dye injection arrangement |
| US9668811B2 (en) | 2010-11-16 | 2017-06-06 | Boston Scientific Scimed, Inc. | Minimally invasive access for renal nerve ablation |
| US9326751B2 (en) | 2010-11-17 | 2016-05-03 | Boston Scientific Scimed, Inc. | Catheter guidance of external energy for renal denervation |
| US9060761B2 (en) | 2010-11-18 | 2015-06-23 | Boston Scientific Scime, Inc. | Catheter-focused magnetic field induced renal nerve ablation |
| US9192435B2 (en) | 2010-11-22 | 2015-11-24 | Boston Scientific Scimed, Inc. | Renal denervation catheter with cooled RF electrode |
| US9023034B2 (en) | 2010-11-22 | 2015-05-05 | Boston Scientific Scimed, Inc. | Renal ablation electrode with force-activatable conduction apparatus |
| US9649156B2 (en) | 2010-12-15 | 2017-05-16 | Boston Scientific Scimed, Inc. | Bipolar off-wall electrode device for renal nerve ablation |
| US9220561B2 (en) | 2011-01-19 | 2015-12-29 | Boston Scientific Scimed, Inc. | Guide-compatible large-electrode catheter for renal nerve ablation with reduced arterial injury |
| US9579030B2 (en) | 2011-07-20 | 2017-02-28 | Boston Scientific Scimed, Inc. | Percutaneous devices and methods to visualize, target and ablate nerves |
| US9186209B2 (en) | 2011-07-22 | 2015-11-17 | Boston Scientific Scimed, Inc. | Nerve modulation system having helical guide |
| US9186210B2 (en) | 2011-10-10 | 2015-11-17 | Boston Scientific Scimed, Inc. | Medical devices including ablation electrodes |
| US10085799B2 (en) | 2011-10-11 | 2018-10-02 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US9420955B2 (en) | 2011-10-11 | 2016-08-23 | Boston Scientific Scimed, Inc. | Intravascular temperature monitoring system and method |
| US9364284B2 (en) | 2011-10-12 | 2016-06-14 | Boston Scientific Scimed, Inc. | Method of making an off-wall spacer cage |
| US9162046B2 (en) | 2011-10-18 | 2015-10-20 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US9079000B2 (en) | 2011-10-18 | 2015-07-14 | Boston Scientific Scimed, Inc. | Integrated crossing balloon catheter |
| US8951251B2 (en) | 2011-11-08 | 2015-02-10 | Boston Scientific Scimed, Inc. | Ostial renal nerve ablation |
| US9119600B2 (en) | 2011-11-15 | 2015-09-01 | Boston Scientific Scimed, Inc. | Device and methods for renal nerve modulation monitoring |
| US9119632B2 (en) | 2011-11-21 | 2015-09-01 | Boston Scientific Scimed, Inc. | Deflectable renal nerve ablation catheter |
| US10856926B2 (en) | 2011-12-09 | 2020-12-08 | Metavention, Inc. | Neuromodulation for metabolic conditions or syndromes |
| US10543034B2 (en) | 2011-12-09 | 2020-01-28 | Metavention, Inc. | Modulation of nerves innervating the liver |
| US10617460B2 (en) | 2011-12-09 | 2020-04-14 | Metavention, Inc. | Neuromodulation for metabolic conditions or syndromes |
| US9265969B2 (en) | 2011-12-21 | 2016-02-23 | Cardiac Pacemakers, Inc. | Methods for modulating cell function |
| US9072902B2 (en) | 2011-12-23 | 2015-07-07 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9028472B2 (en) | 2011-12-23 | 2015-05-12 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9037259B2 (en) | 2011-12-23 | 2015-05-19 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9174050B2 (en) | 2011-12-23 | 2015-11-03 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9186211B2 (en) | 2011-12-23 | 2015-11-17 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9592386B2 (en) | 2011-12-23 | 2017-03-14 | Vessix Vascular, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9402684B2 (en) | 2011-12-23 | 2016-08-02 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9433760B2 (en) | 2011-12-28 | 2016-09-06 | Boston Scientific Scimed, Inc. | Device and methods for nerve modulation using a novel ablation catheter with polymeric ablative elements |
| US9050106B2 (en) | 2011-12-29 | 2015-06-09 | Boston Scientific Scimed, Inc. | Off-wall electrode device and methods for nerve modulation |
| US10660703B2 (en) | 2012-05-08 | 2020-05-26 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices |
| US10321946B2 (en) | 2012-08-24 | 2019-06-18 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices with weeping RF ablation balloons |
| US20140067029A1 (en) * | 2012-08-28 | 2014-03-06 | Boston Scientific Scimed, Inc. | Renal nerve modulation and ablation catheter electrode design |
| US9173696B2 (en) | 2012-09-17 | 2015-11-03 | Boston Scientific Scimed, Inc. | Self-positioning electrode system and method for renal nerve modulation |
| US10398464B2 (en) | 2012-09-21 | 2019-09-03 | Boston Scientific Scimed, Inc. | System for nerve modulation and innocuous thermal gradient nerve block |
| US10549127B2 (en) | 2012-09-21 | 2020-02-04 | Boston Scientific Scimed, Inc. | Self-cooling ultrasound ablation catheter |
| US10835305B2 (en) | 2012-10-10 | 2020-11-17 | Boston Scientific Scimed, Inc. | Renal nerve modulation devices and methods |
| US20150265333A1 (en) * | 2012-10-25 | 2015-09-24 | Kyong-Min Shin | System for ablation utilizing multiple electrodes and method for controlling same |
| US9700369B2 (en) * | 2012-10-25 | 2017-07-11 | Starmed Co., Ltd | System for ablation utilizing multiple electrodes and method for controlling same |
| US9956033B2 (en) | 2013-03-11 | 2018-05-01 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9693821B2 (en) | 2013-03-11 | 2017-07-04 | Boston Scientific Scimed, Inc. | Medical devices for modulating nerves |
| US9808311B2 (en) | 2013-03-13 | 2017-11-07 | Boston Scientific Scimed, Inc. | Deflectable medical devices |
| US9827039B2 (en) | 2013-03-15 | 2017-11-28 | Boston Scientific Scimed, Inc. | Methods and apparatuses for remodeling tissue of or adjacent to a body passage |
| US9297845B2 (en) | 2013-03-15 | 2016-03-29 | Boston Scientific Scimed, Inc. | Medical devices and methods for treatment of hypertension that utilize impedance compensation |
| US10265122B2 (en) | 2013-03-15 | 2019-04-23 | Boston Scientific Scimed, Inc. | Nerve ablation devices and related methods of use |
| US10022182B2 (en) | 2013-06-21 | 2018-07-17 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation having rotatable shafts |
| US9943365B2 (en) | 2013-06-21 | 2018-04-17 | Boston Scientific Scimed, Inc. | Renal denervation balloon catheter with ride along electrode support |
| US9707036B2 (en) | 2013-06-25 | 2017-07-18 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation using localized indifferent electrodes |
| US9833283B2 (en) | 2013-07-01 | 2017-12-05 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US10660698B2 (en) | 2013-07-11 | 2020-05-26 | Boston Scientific Scimed, Inc. | Devices and methods for nerve modulation |
| US10413357B2 (en) | 2013-07-11 | 2019-09-17 | Boston Scientific Scimed, Inc. | Medical device with stretchable electrode assemblies |
| US9925001B2 (en) | 2013-07-19 | 2018-03-27 | Boston Scientific Scimed, Inc. | Spiral bipolar electrode renal denervation balloon |
| US10342609B2 (en) | 2013-07-22 | 2019-07-09 | Boston Scientific Scimed, Inc. | Medical devices for renal nerve ablation |
| US10695124B2 (en) | 2013-07-22 | 2020-06-30 | Boston Scientific Scimed, Inc. | Renal nerve ablation catheter having twist balloon |
| US10722300B2 (en) | 2013-08-22 | 2020-07-28 | Boston Scientific Scimed, Inc. | Flexible circuit having improved adhesion to a renal nerve modulation balloon |
| US9895194B2 (en) | 2013-09-04 | 2018-02-20 | Boston Scientific Scimed, Inc. | Radio frequency (RF) balloon catheter having flushing and cooling capability |
| US10952790B2 (en) | 2013-09-13 | 2021-03-23 | Boston Scientific Scimed, Inc. | Ablation balloon with vapor deposited cover layer |
| US11246654B2 (en) | 2013-10-14 | 2022-02-15 | Boston Scientific Scimed, Inc. | Flexible renal nerve ablation devices and related methods of use and manufacture |
| US9687166B2 (en) | 2013-10-14 | 2017-06-27 | Boston Scientific Scimed, Inc. | High resolution cardiac mapping electrode array catheter |
| US9962223B2 (en) | 2013-10-15 | 2018-05-08 | Boston Scientific Scimed, Inc. | Medical device balloon |
| US9770606B2 (en) | 2013-10-15 | 2017-09-26 | Boston Scientific Scimed, Inc. | Ultrasound ablation catheter with cooling infusion and centering basket |
| US10945786B2 (en) | 2013-10-18 | 2021-03-16 | Boston Scientific Scimed, Inc. | Balloon catheters with flexible conducting wires and related methods of use and manufacture |
| US10271898B2 (en) | 2013-10-25 | 2019-04-30 | Boston Scientific Scimed, Inc. | Embedded thermocouple in denervation flex circuit |
| US11202671B2 (en) | 2014-01-06 | 2021-12-21 | Boston Scientific Scimed, Inc. | Tear resistant flex circuit assembly |
| US11000679B2 (en) | 2014-02-04 | 2021-05-11 | Boston Scientific Scimed, Inc. | Balloon protection and rewrapping devices and related methods of use |
| US9907609B2 (en) | 2014-02-04 | 2018-03-06 | Boston Scientific Scimed, Inc. | Alternative placement of thermal sensors on bipolar electrode |
| US11160569B2 (en) | 2015-09-17 | 2021-11-02 | Texas Heart Institute | Esophageal probes and methods |
| EP3349640A4 (en) * | 2015-09-17 | 2019-05-15 | Baylor College of Medicine | Esophageal probes and methods |
| AU2016324501B2 (en) * | 2015-09-17 | 2021-07-08 | Baylor College Of Medicine | Esophageal probes and methods |
| CN108430301A (en) * | 2015-09-17 | 2018-08-21 | 贝勒医学院 | Oesophagus probe and method |
| CN108430301B (en) * | 2015-09-17 | 2022-01-25 | 贝勒医学院 | Esophageal probes and methods |
| WO2017049313A1 (en) * | 2015-09-17 | 2017-03-23 | Baylor College Of Medicine | Esophageal probes and methods |
| US10524859B2 (en) | 2016-06-07 | 2020-01-07 | Metavention, Inc. | Therapeutic tissue modulation devices and methods |
Also Published As
| Publication number | Publication date |
|---|---|
| US20020161361A1 (en) | 2002-10-31 |
| WO2002047565A2 (en) | 2002-06-20 |
| US6558378B2 (en) | 2003-05-06 |
| WO2002047565A3 (en) | 2002-08-22 |
| AU2002230812A1 (en) | 2002-06-24 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US6558378B2 (en) | RF ablation system and method having automatic temperature control | |
| US6171305B1 (en) | RF ablation apparatus and method having high output impedance drivers | |
| US6488678B2 (en) | RF ablation apparatus and method using unipolar and bipolar techniques | |
| US6050994A (en) | RF ablation apparatus and method using controllable duty cycle with alternate phasing | |
| US6049737A (en) | Catheter having common lead for electrode and sensor | |
| US6635056B2 (en) | RF ablation apparatus and method using amplitude control | |
| US6666862B2 (en) | Radio frequency ablation system and method linking energy delivery with fluid flow | |
| US6730078B2 (en) | RF ablation apparatus and method using multi-frequency energy delivery | |
| US6042580A (en) | Electrode having composition-matched, common-lead thermocouple wire for providing multiple temperature-sensitive junctions | |
| US6045550A (en) | Electrode having non-joined thermocouple for providing multiple temperature-sensitive junctions | |
| US6761716B2 (en) | System and method for assessing electrode-tissue contact and lesion quality during RF ablation by measurement of conduction time | |
| US20030187430A1 (en) | System and method for measuring power at tissue during RF ablation | |
| US6740080B2 (en) | Ablation system with selectable current path means | |
| US6391024B1 (en) | RF ablation apparatus and method having electrode/tissue contact assessment scheme and electrocardiogram filtering |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |