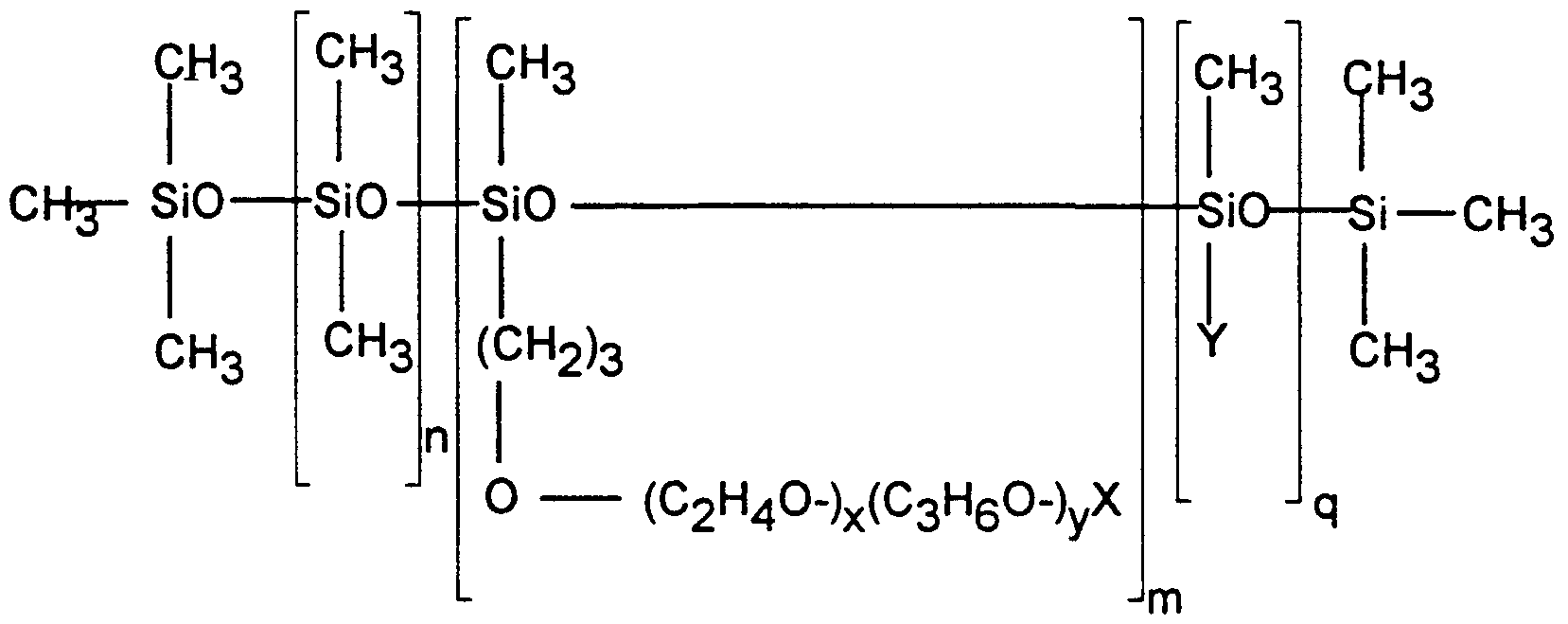WO1997002011A1 - Oral compositions - Google Patents
Oral compositions Download PDFInfo
- Publication number
- WO1997002011A1 WO1997002011A1 PCT/US1996/010815 US9610815W WO9702011A1 WO 1997002011 A1 WO1997002011 A1 WO 1997002011A1 US 9610815 W US9610815 W US 9610815W WO 9702011 A1 WO9702011 A1 WO 9702011A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- ofthe
- composition according
- weight
- aminoalkylsilicone
- alkyl
- Prior art date
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/89—Polysiloxanes
- A61K8/891—Polysiloxanes saturated, e.g. dimethicone, phenyl trimethicone, C24-C28 methicone or stearyl dimethicone
- A61K8/894—Polysiloxanes saturated, e.g. dimethicone, phenyl trimethicone, C24-C28 methicone or stearyl dimethicone modified by a polyoxyalkylene group, e.g. cetyl dimethicone copolyol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/02—Cosmetics or similar toiletry preparations characterised by special physical form
- A61K8/0216—Solid or semisolid forms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/347—Phenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/817—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a single or double bond to nitrogen or by a heterocyclic ring containing nitrogen; Compositions or derivatives of such polymers, e.g. vinylimidazol, vinylcaprolactame, allylamines (Polyquaternium 6)
- A61K8/8176—Homopolymers of N-vinyl-pyrrolidones. Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/86—Polyethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/84—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions otherwise than those involving only carbon-carbon unsaturated bonds
- A61K8/89—Polysiloxanes
- A61K8/896—Polysiloxanes containing atoms other than silicon, carbon, oxygen and hydrogen, e.g. dimethicone copolyol phosphate
- A61K8/898—Polysiloxanes containing atoms other than silicon, carbon, oxygen and hydrogen, e.g. dimethicone copolyol phosphate containing nitrogen, e.g. amodimethicone, trimethyl silyl amodimethicone or dimethicone propyl PG-betaine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q11/00—Preparations for care of the teeth, of the oral cavity or of dentures; Dentifrices, e.g. toothpastes; Mouth rinses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q11/00—Preparations for care of the teeth, of the oral cavity or of dentures; Dentifrices, e.g. toothpastes; Mouth rinses
- A61Q11/02—Preparations for deodorising, bleaching or disinfecting dentures
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/20—Chemical, physico-chemical or functional or structural properties of the composition as a whole
- A61K2800/22—Gas releasing
- A61K2800/222—Effervescent
Definitions
- the present invention relates to oral compositions such as toothpastes, toothpowders, liquid dentifrices, mouthwashes, denture cleansers, chewing gums, candies and the like.
- oral compositions having enhanced antiplaque activity together with excellent cleansing performance, physical characteristics, and in-use performance characteristics.
- Plaque is initiated when bacteria adhered to pellicle form a proteinaceous film on the surface of teeth.
- the adherent bacteria metabolise dietary constituents and reproduce and aggregate to form the tenacious deposit known as plaque.
- Plaque generally consists of bacteria, bacterial end products such as polysaccharides, inorganic salts and salivary proteins. Plaque bacteria ferment dietary carbohydrates to organic acids which demineralise enamel resulting in tooth decay.
- Calculus is essentially plaque that has been mineralised with calcium phosphates salts. As calculus matures and hardens, it tends to stain noticeably due to adsorption of dietary chromagens. In addition to their unattractive appearance, calculus deposits at the gum line are a contributing source of gingivitis and periodontal disease. Besides the hygienic and health problems resulting from plaque, research has shown that the primary source of bad breath is the retention and subsequent degradation of dead cellular material sloughed off continuously by the normal, healthy mouth.
- Modern dental hygiene and denture preparations typically contain antiplaque and/or antitartar agents, as well as antimicrobial agents and flavorants.
- Antimicrobial action could affect plaque formation by either reducing the number of bacteria in the mouth/dentures or by killing those bacteria trapped in the film to prevent further growth and metabolism.
- Flavorants may alleviate the problem of bad breath via a deodorising action.
- Some antimicrobial agents, e.g. menthol may, also serve as breath deodorisers.
- the efficacy of antimicrobial agents depends largely on their intraoral/denture retention, particularly their retention on the surface ofthe teeth or dentures where plaque is formed.
- a typical disadvantage of known dental preparations is that only a relatively short time during which the teeth are being cleaned or the mouth is being rinsed is available for antimicrobial agents in the preparations to take effect.
- the problem is compounded by the fact that dentifrice preparations are used infrequently: most are used once or, perhaps, twice daily. Consequently, the long time period between brushings for a majority ofthe population provides optimum plaque forming conditions.
- Oral compositions if not aqueous themselves, are required to act within an aqueous environment. Many ingredients of such compositions act more effectively if they can first be effectively dispersed within an aqueous medium. Once dispersed, those ingredients which act by depositing on the teeth or other surfaces within, or for use within, the mouth, are then required to be deposited from aqueous dispersion in effective amounts.
- GB-A-689,679 discloses a mouthwash containing an organopolysiloxane for preventing adhesion of, or for removing tars, stains, tartar and food particles from the teeth.
- the mouthwash may include antiseptic compounds, such as thymol, and flavouring and perfuming agents.
- US-A-2,806,814 discloses dental preparations including, in combination, a higher aUphatic acyl amide of an amino carboxylic acid compound as an active and a silicone compound.
- silicone compounds have been proposed for prevention of adhesion or to facilitate the removal of tars, stains, tartar and the like from teeth.
- the silicone compound is said to act as a synergist in improving the antibacterial and acid inhibiting activity ofthe active ingredient.
- Dimethyl polysiloxanes are said to be particularly effective.
- Flavouring oils and/or menthol may be included.
- US-A-3624120 discloses quaternary ammomum salts of cyclic siloxane polymers for use as cationic surfactants, bactericides and as anticariogenic agents.
- EP-A-376,363 discloses dentifrices containing amino alkyl silicones and sarcosinate surfactants which form a hydrophobic barrier on the surface of teeth.
- the present invention provides oral compositions, comprising an aminoalkylsilicone, having improved efficacy on plaque, mucilaginous and bacterial deposits and which further comprise a surface active agent chosen to enhance the dispersion ofthe aminoalkylsilicone, whilst at the same time providing improved substantivity and efficacy on teeth, gums and dentures; together with excellent physical characteristics, and in-use performance characteristics.
- an oral composition in the form of a toothpaste, powder, liquid dentifrice, mouthwash, denture cleanser, chewing gum or candy comprising one or more oral composition components selected from abrasives, binders, humectants, surfactants, fluoride ion sources, anti-calculus agents and sweeteners and additionally comprising: i) an aminoalkylsilicone; and ii) a silicone surfactant having the general formula (I)
- X is selected from hydrogen, alkyl, alkoxy and acyl groups having from about 1 to about 16 carbon atoms
- Y is CH3
- q is 0, n is from about 1 to about 100, m is from about 1 to about 40, the molecular weight ofthe residue is from about 50 to about 2000, and x and - 4 - y are such that the weight ratio of oxyethylene:oxypropylene is from about 100:0 to about 0:100.
- compositions ofthe invention thus comprise an aminoalkylsilicone antiplaque agent and a dimethicone copolyol surfactant, while preferred compositions additionally comprise a lipophilic compound and/or one or more oral composition components selected from abrasives, binders, humectants, surfactants, fluoride ion sources, anti-calculus agents and sweeteners. Each of these will be discussed in turn.
- aminoalkylsilicone is selected from noncyclic, hydro ⁇ phobic aminoalkysilicones having a formula comprising two basic units:
- Rl and R ⁇ are independently selected from H ,alkyl and alkenyl of about 1 to about 10 carbons optionally substituted with fluoro or cyano groups, hydroxy, alkoxy, and acetoxy, for example, wherein Rl and R2 are independently selected from methyl, ethyl, phenyl, vinyl, trifluoropropyl and cyanopropyl, and R is
- R ⁇ is a divalent alkylene of about 1-20, preferably about 3-5 carbon atoms optionally substituted or interrupted by O atoms
- R 4 , R5 and R6 which may be the same or different are selected from H, alkyl of about 1-20, preferably about 1-10, more preferably about 1-4 carbons optionally substituted or interrupted by N and/or O atoms
- X" is a monovalent anion such as halide, hydroxide, and tosylate, said aminoalkylsilicone including about 60% or less, preferably from about 0.1-30%, more preferably from 0.2-10% and especially from about 0.5-2% of unit (1) on a repeating unit basis.
- aminoalkylsilicones comprise amo ⁇ dimethicones.
- Amodimethicones are polydimethylsiloxane polymers containing aminoalkyl groups.
- the aminoalkyl groups may be present either pendant or at one or more ends ofthe polydimethylsiloxane chain.
- aminoalkylsilicones in which aminoalkyl moiety R is selected from (CH 2 )3NH 2 , (CH 2 )3NHCH2CH2NH2, (CH 2 )3N(CH 2 CH2 ⁇ H)2, (CH2)3NH3 + X-, and (CH2)3N(CH 3 ) 2 (Ci8H37) + X-, and especially from (CH2)3NH2 and (CH2)3 HCH2CH2NH2.
- aminoalkyl silicones having an average molecular weight of about 5,000 and above, preferably from about 5000 to about 100,000, more preferably from about 5000 to about 30,000.
- Aminoalkylsilicone compounds suitable for use herein are well known. Methods of preparing aminoalkylsilicones are given in, for example, US-A- 2,930,809.
- amodimethicones examples include OSI's Magnasoft fluid. These polymers comprise aminoalkyl groups affixed to a predominantly polydimethylsiloxane structure. The typical structure of Magnasoft's aminoalkyl group-containing units is
- the aminoalkylsilicone is generally present in a level offrom about 0.01% to about 25%, preferably from about 0.1% to about 5%, more preferably from about 0.5% to about 1.5% by weight.
- a second essential ingredient ofthe oral compositions ofthe invention is a silicone surfactant having the general formula (I)
- X is selected from hydrogen, alkyl, alkoxy and acyl groups having from about 1 to about 16 carbon atoms, Y is CH3, q is 0, n is from about 1 to about 100, m is from about 1 to about 40, the molecular weight ofthe residue (C 2 H4 ⁇ -) ⁇ (C3H6 ⁇ -)yX is from about 50 to about 2000, and x and y are such that the weight ratio of oxyethylene:oxypropylene is from about 100:0 to about 0:100.
- the silicone surfactant assists in dispersing the aminoalkylsilicone antiplaque agent in aqueous media whilst still allowing the antiplaque agent to deposit onto surfaces such as teeth, gums or artificial dentures.
- Suitable dimethicone copolyols are those marketed under the Trade Name Silwet by Union Carbide.
- the silicone surfactant is generally present in a level offrom about 0.01% to about 25%, preferably from about 0.3% to about 10%, more preferably from about 0.5% to about 5% by weight. It will be appreciated that the precise amount will depend on the amount ofthe aminoalkylsilicone antiplaque agent used. In general the ratio of silicone surfactant to aminoalkylsilicone antiplaque agent will be from about 0.5:1 to 15:1, more preferably from about 1:1 to 10:1, most preferably from about 2:1 to 8:1 by weight.
- the oral compositions ofthe invention preferably also include a lipophilic compound.
- lipophilic compounds suitable for use herein are oil-like materials which are soluble or solubilisable in the aminoalkylsilicone, preferably at a level of at least about 1%, more preferably at least about 5% by weight at 25°C.
- Prefe ⁇ ed lipophilic compounds are selected from flavorants, physiological cooling agents and antimicrobial compounds.
- the aminoalkyl ⁇ silicone acts to enhance the substantivity ofthe lipophilic compound to teeth and/or dentures, thereby providing enhanced and/or sustained flavour impact and antimicrobial efficacy.
- Lipophilic flavorants suitable for use herein comprise one or more flavour components selected from wintergreen oil, oregano oil, bay leaf oil, peppermint oil, spearmint oil, clove oil, sage oil, sassafras oil, lemon oil, orange oil, anise oil, benzaldehyde, bitter almond oil, camphor, cedar leaf oil, marjoram oil, citronella oil, lavender oil, mustard oil, pine oil, pine needle oil, rosemary oil, thyme oil, cinnamon leaf oil, and mixtures thereof.
- Lipophilic antimicrobial compounds suitable for use herein include thymol, menthol, triclosan, 4-hexylresorcinol, phenol, eucalyptol, benzoic acid, benzoyl peroxide, butyl paraben, methyl paraben, propyl paraben, salicylamides, and mixtures thereof.
- Physiological cooling agent suitable for use herein include carboxamides, menthane esters and menthane ethers, and mixtures thereof.
- Suitable menthane ethers for use herein are selected from those with the formula:
- R5 is an optionally hydroxy substituted aliphatic radical containing up to 25 carbon atoms, preferably up to 5 carbon atoms, and where X is hydrogen or hydroxy, such as those commercially available under the trade name Takasago, from Takasago International Co ⁇ oration.
- a particularly prefe ⁇ ed cooling agent for use in the compositions ofthe present invention is Takasago 10 [3-1- menthoxy propan-l,2-diol (MPD)].
- MPD is a monoglycerine derivative of 1- menthol and has excellent cooling activity.
- the level of lipophilic compound in the compositions ofthe invention is generally in the range from about 0.01% to about 10%, preferably from about 0.05% to about 5%, more preferably from about 0.1% to about 3% by weight.
- compositions in the form of toothpastes, denture cleansing liquids and pastes and the like will generally comprise a binder or thickening agent.
- Binders suitable for use herein include carboxyvinyl polymers, ca ⁇ ageenan, hydroxy ⁇ ethyl cellulose and water soluble salts of cellulose ethers such as sodium carboxymethyl cellulose and sodium carboxymethyl hydroxyethyl cellulose.
- Natural gums such as gum karaya, xanthan gum, gum arabic, and gum tragacanth can also be used.
- Colloidal magnesium aluminium silicate or finely divided silica can be used as part ofthe thickening agent to further improve texture.
- Binders/thickening agents can be used in an amount from about 0.1% to about 5.0%, preferably from about 0.1 to about 1% by weight ofthe total composition.
- humectant material in a toothpaste to keep the composition from hardening upon exposure to air.
- Certain humectants can also impart a desirable sweetness to toothpaste compositions.
- SUBSTTTUTE SHEET (RULE 26) and mouthwashes can also contain a quantity of humectant.
- Suitable humectants include glycerine, sorbitol, xylitol, polyethylene glycols, propylene glycol, other edible polyhydric alcohols, and mixtures thereof. When present, humectants generally represent from about 10% to about 70%, by weight ofthe compositions ofthe invention.
- liquid dentifrices and denture cleansers in liquid or paste fo ⁇ n will generally comprise an abrasive polishing material.
- the abrasive polishing material contemplated for use herein can be any material which does not excessively abrade dentine or denture acrylic.
- silicas including xerogels, hydrogels, aerogels and precipitates, calcium and magnesium carbonates, calcium ortho-, pyro- meta- and polyphosphates such as dicalcium orthophosphate dihydrate, calcium pyrophosphate, tricalcium phosphate, and calcium polymetaphosphate, insoluble sodium polymetaphosphate, alumina and hydrates thereof such as alpha alumina trihydrate, aluminosilicates such as calcined aluminium silicate and aluminium silicate, magnesium and zirconium silicates such as magnesium trisilicate and thermosetting polymerised resins such as particulate condensation products of urea and formaldehyde, polymethylmethacrylate, powdered polyethylene and others such as disclosed in US-A-3, 070,510, December 25, 1962. Mixtures of abrasives can also be used.
- the abrasive polishing materials generally have an average particle size offrom about 0.1 to about 30 microns, preferably from about 5 to 15 micron
- Silica dental abrasives of various types offer exceptional dental cleaning and polishing performance without unduly abrading tooth enamel or dentine.
- the silica abrasive can be precipitated silica or silica gels such as the silica xerogels described in Pader et al., US-A-3,538,230, issued March 2, 1970 and DiGiulio, US-A-3,862,307, June 21, 1975, for example silica xerogels marketed under the tradename "Syloid" by W. R. Grace & Company, Davison Chemical Division.
- Suitable precipitated silica materials include those marketed by the J. M. Huber Corporation under the tradename, "Zeodent", particularly the silica carrying the designation "Zeodent 119". These silica abrasives are described in US-A-4,340,583, July 29, 1982.
- the abrasive is generally present in dentifrice formulations ofthe invention at a level offrom about 10% to about 70%, preferably from about 15% to about 25% by weight.
- compositions can also contain surfactants.
- Suitable surfactants are those which are reasonably stable and foam throughout a wide pH range, including non-soap anionic, nonionic, cationic, zwitterionic and amphoteric organic synthetic detergents. Many ofthese suitable agents are disclosed by Gieske et al. in US-A-4,051,234, September 27, 1977.
- Suitable surfactants include alkyl sulphates; condensation products of ethylene oxide with fatty acids, fatty alcohols, fatty amides, polyhydric alcohols (e.g. sorbitan monostearate, sorbitan oleate), alkyl phenols (e.g. Tergitol) and polypropyleneoxide or polyoxybutylene (e.g.
- amine oxides such as dimethyl cocamine oxide, dimethyl lauryl amine oxide and cocoalkyldimethyl amine oxide (Aromox); polysorbates such as Tween 40 and Tween 80 (Hercules); sorbitan stearates, sorbitan monooleate, etc.; sarcosinates such as sodium cocoylsarcosinate, sodium lauroyl sarcosinate (Hamposyl-95 ex W. R.
- cationic surfactants such as cetyl pyridinium chloride, cetyl trimethyl ammomum bromide, di-isobutyl phenoxy ethoxy ethyl- dimethyl benzyl ammonium chloride and coconut alkyl trimethyl ammomum nitrate
- a soluble fluoride ion source can also be inco ⁇ orated in the present compositions.
- the soluble fluoride ion source is used in amounts sufficient to provide from about 50 to about 3500 ppm ofthe fluoride ion.
- Prefe ⁇ ed fluorides are sodium fluoride, stannous fluoride, indium fluoride, zinc ammomum fluoride, tin ammonium fluoride, calcium fluoride and sodium monofluorophosphate.
- Norris et al., US-A-2,946,735, issued July 26, 1960 and Widder et al., US-A-3,678,154, issued July 18, 1972 disclose such salts as well as others.
- compositions can also include an anti-calculus agent.
- Suitable anti-calculus agents include the di- and tetra-alkali metal pyrophosphates as set out in EP-A-097476.
- Specific salts include tetra alkali metal pyrophosphate, dialkali metal diacid pyrophosphate, trialkali metal monoacid pyrophosphate and mixtures thereof, wherein the alkali metals are sodium or potassium.
- the salts are useful in both their hydrated and unhydrated forms.
- the amount of pyrophosphate salt useful in these compositions is any effective amount and is generally enough to provide in composition at least 1.0% P 2 ⁇ 7" 4 , preferably
- SUBS ⁇ iUIE SHEET 10 - from about 1.5% to about 10%, more preferably from about 3% to about 6% by weight of composition.
- the pyrophosphate salts are described in more detail in Kirk & Othmer, Encyclopedia of Chemical Technologv. Second Edition, Volume 15, Interscience Publishers (1968).
- Zinc salts are disclosed in US-A-4, 100,269, US-A-4,416,867, US-A-4,425,325 and US-A- 4,339,432.
- a prefe ⁇ ed agent ofthe zinc variety is zinc citrate.
- Zinc compounds can be present in amounts sufficient to provide from about 0.01% to about 4%, preferably from about 0.05% to about 1% by weight of zinc ion.
- Suitable anti-calculus agents include the synthetic anionic polymers (including polyacrylates and copolymers of maleic anhydride or acid and methyl vinyl ether (e.g. Gantrez) as described in US-A-4,627,977, polyamino propane sulphonic acid, polyphosphates (e.g. tripolyphosphate, hexameta ⁇ phosphate), diphosphonates (e.g. EHDP, AHP), polypeptides (e.g. poly ⁇ aspartic and polyglutamic acids), and mixtures thereof.
- synthetic anionic polymers including polyacrylates and copolymers of maleic anhydride or acid and methyl vinyl ether (e.g. Gantrez) as described in US-A-4,627,977, polyamino propane sulphonic acid, polyphosphates (e.g. tripolyphosphate, hexameta ⁇ phosphate), diphosphonates (e.g. EHDP, AHP), polypeptides ( e
- Sweetening agents which can be used include aspartame, acesulphame, saccharin, dextrose, levulose and sodium cyclamate. Sweetening agents are generally used at levels offrom about 0.005% to about 2% by weight of composition.
- water-soluble antibacterial agents such as chlorhexidine digluconate, quaternary ammonium antibacterial compounds and water-soluble sources of certain metal ions such as zinc, copper, silver and stannous (e.g., zinc, copper and stannous chloride, and silver nitrate); pigments such as titanium dioxide; orally acceptable dyes/colorants such as FD&C Blue #1, FD&C Yellow #10, FD&C Red #40; antioxidants, vitamins such as vitamin C and E, other antiplaque agents such as stannous salts, copper salts, strontium salts and magnesium salts; pH adjusting agents, anticaries agents such as urea, calcium glycerophosphate, sodium trimetaphosphate, plant extracts, desensitising agents for sensitive teeth such as potassium nitrate and potassium citrate, and mixtures thereof.
- water-soluble antibacterial agents such as chlorhexidine digluconate, quaternary ammonium antibacterial compounds and water-soluble sources of certain metal ions such as zinc,
- mouthwashes comprise a water/alcohol solution, flavour, humectant, sweetener, sudsing agent, and colorant as described above.
- Mouthwashes can include ethanol at a level offrom 0 to 60%, preferably from 5 to 30% by weight.
- Denture cleanser compositions ofthe invention can additionally include one or more bleaching agents, organic peroxyacid precursors, effervescence generators, chelating agents, etc.
- the bleaching agent takes the form of an inorganic persalt and can be selected from any ofthe well-known bleaching agents known for use in denture cleansers such as the alkali metal and ammomum persulphates, perborates, percarbonates and pe ⁇ hosphates and the alkali metal and alkaline earth metal peroxides.
- suitable bleaching agents include potassium, ammonium, sodium and lithium persulphates and perborate mono- and tetrahydrates, sodium pyrophosphate peroxyhydrate and magnesium, calcium, strontium and zinc peroxides.
- the alkali metal persulphates, perborates and mixtures thereof are prefe ⁇ ed for use herein, highly prefe ⁇ ed being the alkali metal perborates.
- the tablet compositions herein will provide excellent antimicrobial activity even in the absence of alkali metal persulphates.
- the amount of bleaching agent in the total composition is generally from about 5 to about 70% preferably from about 10% to about 50%.
- the overall persulphate:perborate ratio is suitably from about 5:1 to about 1:5, more especially from about 2:1 to about 1:2.
- the denture cleansing compositions can also inco ⁇ orate an effervescence generator, i.e. a material which in the presence of water releases carbon dioxide or oxygen with effervescence.
- the effervescence generator can be selected from generators which are effective under acid, neutral or alkaline pH conditions, but preferably it consists of a combination of a generator which is effective or most effective under acid or neutral pH conditions and a generator which is effective or most effective under alkaline pH conditions.
- Effervescence generators which are effective under acid or neutral pH conditions include a combination of at least one alkali metal carbonate or bicarbonate, such as sodium bicarbonate, sodium carbonate, sodium sesquicarbonate, potassium carbonate, potassium bicarbonate, or mixtures thereof, in admixture with at least one non-toxic, physiologically-acceptable organic acid, such as tartaric, fumaric, citric, malic, maleic, gluconic, succinic, salicylic, adipic or sulphamic acid, sodium fumarate, sodium or potassium acid phosphates, betaine hydrochloride or mixtures thereof. Ofthese, malic acid is prefe ⁇ ed.
- Effervescence generators which are effective under alkaline pH conditions include persalts such as alkali and alkaline earth metal peroxoborates as well as perborates, persulphates, percarbonates, pe ⁇ hosphates and mixtures thereof as previously described, for example, a mixture of an alkali metal perborate (anhydrous, mono- or tetrahydrate) with a monopersulphate such as Caroat R marketed by E I du Point de Nemours Co. and which is a 2:1:1 mixture of monopersulphate, potassium sulphate and potassium bisulphate and which has an active oxygen content of about 4.5%.
- persalts such as alkali and alkaline earth metal peroxoborates as well as perborates, persulphates, percarbonates, pe ⁇ hosphates and mixtures thereof as previously described, for example, a mixture of an alkali metal perborate (anhydrous, mono- or tetrahydrate) with a monopersulphate such as Caroat R marketed by
- the effervescence generator takes the form of a solid base material which in the presence of water releases carbon dioxide or oxygen with effervescence.
- the solid base material inco ⁇ orates a (bi)carbonate/acid effervescent couple optionally in combination with a perborate/persulphate oxygen effervescence generator.
- the combination of generators is valuable for achieving optimum dissolution characteristics and pH conditions for achieving optimum cleaning and antimicrobial activity.
- the (bi)carbonate components generally comprise from about 5% to about 65%, preferably from about 25% to 55% ofthe total composition; the acid components generally comprise from about 5% to about 50%, preferably from about 10% to about 30% ofthe total composition.
- the denture cleansing compositions ofthe invention can be supplemented by other known components ofsuch formulations.
- An especially prefe ⁇ ed additional component is an organic peroxyacid precursor, which in general terms can be defined as a compound having a titre of at least 1.5ml of 0.1N sodium thiosulphate in the following peracid formation test.
- a test solution is prepared by dissolving the following materials in 1000 mis distilled water: sodium pyrophosphate (Na4P 2 ⁇ 7.10H2 ⁇ ) 2.5g sodium perborate (NaB0 2 .H 2 0 2 .3H 2 0) having 10.4% available oxygen 0.615g sodium dodecylbenzene sulphonate 0.5g
- the mixture obtained by addition ofthe activator is vigorously stirred and maintained at 60°C. After 5 minutes from addition, a 100 ml portion ofthe solution is withdrawn and immediately pipetted onto a mixture of 250 g cracked ice and 15 ml glacial acetic acid. Potassium iodide (0.4 g) is then added and the liberated iodine is immediately titrated with 0.1 N sodium thiosulphate with starch as indicator until the first disappearance ofthe blue colour. The amount of sodium thiosulphate solution used in ml is the titre of the bleach activator.
- the organic peracid precursors are typically compounds containing one or more acyl groups, which are susceptible to perhydrolysis.
- the prefe ⁇ ed activators are those ofthe N-acyl or O-acyl compound type containing a acyl radical R-CO wherein R is a hydrocarbon or substituted hydrocarbon group having preferably from about 1 to about 20 carbon atoms.
- suitable peracid precursors include:
- TAED N.NjN'N' -tetraacetylethylenediamine
- TAHD co ⁇ esponding hexamethylenediamine
- Examples of compounds of this type include phenyl acetate, sodium acetoxy benzene sulphonate, trichloroethylacetate, sorbitol hexaacetate, fructose pentaacetate, p-nitrobenzaldehyde diacetate, isopropenyl acetate, acetyl aceto hydroxamic acid, and acetyl salicylic acid.
- esters of a phenol or substituted phenol with an alpha-chlorinated lower aliphatic carboxylic acid such as chloroacetylphenol and chloroacetylsalicylic acid, as disclosed in US-A- 3,130,165.
- Prefe ⁇ ed compoimds of this type are those wherein: a) Ac is R3-CO and R3 is a linear or branched alkyl group containing from 6 to 20, preferably 6 to 12, more preferably 7 to 9 carbon atoms and wherein the longest linear alkyl chain extending from and including the carbonyl carbon contains from 5 to 18, preferably 5 to 10 carbon atoms, R3 optionally being substituted (preferably alpha to the carbonyl moiety) by Cl, Br, OCH3 or OC 2 H5.
- Examples of this class of material include sodium 3,5,5-trimethylhexanoyloxybenzene sulphonate, sodium 3,5,5-trimethylhexanoyloxybenzoate, sodium 2- ethylhexanoyl oxybenzenesulphonate, sodium nonanoyl oxybenzene sulphonate and sodium octanoyl oxybenzenesulphonate, the acyloxy group in each instance preferably being p-substituted; b) Ac has the formula R3(AO) m XA wherein R3 is a linear or branched alkyl or alkylaryl group containing from 6 to 20, preferably from 6 to 15 carbon atoms in the alkyl moiety, R5 being optionally substituted by Cl, Br, OCH3, or OC 2 H5, AO is oxyethylene or oxypropylene, m is from 0 to 100, X is O, NR4 or CO-NR4, and A is CO, CO-CO
- m is preferably from 0 to 10
- R3 is preferably C -C ⁇ 2, more preferably C6-C10 alkyl when m is zero and C9-C15 when m is non ⁇ zero.
- the leaving group L is as defined above. 5) Acyl-cyanurates, such as triacetyl- or tribenzoylcyanurates, as disclosed in US patent specification No. 3,332,882.
- Optionally substituted anhydrides of benzoic or phthalic acid for example, benzoic anhydride, m-chlorobenzoic anhydride and phthalic anhydride.
- prefe ⁇ ed are organic peracid precursors of types 1(c) and 4(a).
- the level of peroxyacid bleach precursor by weight ofthe total composition is preferably from about 0.1% to about 10%, more preferably from about 0.5% to about 5% and is generally added in the form of a bleach precursor agglomerate.
- the bleach precursor agglomerates prefe ⁇ ed for use herein generally comprise a binder or agglomerating agent in a level offrom about 5% to about 40%, more especially from about 10% to about 30% by weight thereof.
- Suitable agglomerating agents include polyvinylpyrrolidone, poly(oxyethylene) of molecular weight 20,000 to 500,000, polyethyleneglycols of molecular weight offrom about 1000 to about 50,000, Carbowax having a molecular weight of from 4000 to 20,000, nonionic surfactants, fatty acids, sodium carboxymethyl cellulose, gelatine, fatty alcohols, phosphates and polyphosphates, clays, aluminosilicates and polymeric polycarboxylates.
- polyethyleneglycols are highly prefe ⁇ ed, especially those having molecular weight offrom about 1,000 to about 30,000, preferably 2000 to about 10,000.
- bleach precursor agglomerates which comprise from about 10% to about 75%, preferably from about 20% to about 60% by weight thereof of peroxyacid bleach precursor, from about 5% to about 60% preferably from about 5% to about 50%, more preferably from about 10% to about 40% of a (bi) carbonate/acid effervescent couple, from about 0% to about 20% of a peroxoborate, and from about 5% to about 40%, preferably from about 10% to about 30% of an agglomerating agent.
- the final bleach precursor granules desirably have an average particle size offrom about 500 to about 1500, preferably from about 500 to about 1,000 wm, this being valuable from the viewpoint of optimum dissolution performance and aesthetics.
- the level of bleach precursor agglomerates is preferably from about 1% to about 20%, more preferably from about 5% to about 15% by weight of composition.
- the denture cleansing compositions ofthe invention can be in paste, tablet, granular or powder form, although tablet-form compositions are highly prefe ⁇ ed herein. Compositions in tablet form can be single or multiple layered tablets.
- Denture cleansing compositions ofthe invention can be supplemented by other usual components of such formulations, especially surfactants, chelating agents, enzymes, flavorants, physiological cooling agents, antimicrobial compounds, dyestuffs, sweeteners, tablet binders and fillers, foam depressants such as dimethylpolysiloxanes, foam stabilisers such as the fatty acid sugar esters, preservatives, lubricants such as talc, magnesium stearate, finely divided amo ⁇ hous pyrogenic silicas, etc.
- the free moisture content ofthe final composition is desirably less than about 1% and especially less than about 0.5%.
- Tablet binders and fillers suitable for use herein include polyvinylpynolidone, poly (oxyethylene) of molecular weight 20,000 to 500,000, polyethyleneglycols of molecular weight offrom about 1000 to about 50,000, Carbowax having a molecular weight offrom 4000 to 20,000, nonionic surfactants, fatty acids, sodium carboxymethyl cellulose, gelatine, fatty alcohols, clays, polymeric polycarboxylates, sodium carbonate, calcium carbonate, calcium hydroxide, magnesium oxide, magnesium hydroxide carbonate, sodium sulphate, proteins, cellulose ethers, cellulose esters, polyvinyl alcohol, alginic acid esters, vegetable fatty materials of a pseudocolloidal character.
- polyethyleneglycols are highly preferred, especially those having molecular weight offrom about 1,000 to about 30,000, preferably from about 12,000 to about 30,000.
- the surface active agent used in the denture cleansing compositions ofthe invention can be selected from the many available that are compatible with the other ingredients ofthe denture cleanser, both in the dry state and in solution. Such materials are believed to improve the effectiveness ofthe other ingredients ofthe composition by aiding their penetration into the interdental surfaces. Also, these materials aid in the removal of food debris attached to the teeth.
- a dry powder or granular anionic surface active agent such as sodium lauryl sulphate, sodium N-lauroylsarcosinate, sodium lauryl sulphoacetate or dioctyl sodium sulphosuccinate or ricinoleyl sodium sulphosuccinate, may, for example, be included in the composition and preferably the surface active agent comprises between 0.5 and 4 percent ofthe composition.
- Suitable cationic, non-ionic and ampholytic surface active agents include, for example, quaternary ammomum compounds such as cetyltrimethylammomum bromide, condensation products of alkylene oxides such as ethylene or propylene oxide with fatty alcohols, phenols, fatty amines or fatty acid alkanolamides, the fatty acid alkanolamides themselves, esters of long-chained (C8-C 22 ) fatty acids with polyalcohols or sugars, for example glyceryl- monostearate or saccharose monolaurate or sorbitolpolyoxyethylenemono-or di-stearate, betaines, sulphobetaines or long-chain alkylaminocarboxylic acids.
- quaternary ammomum compounds such as cetyltrimethylammomum bromide
- condensation products of alkylene oxides such as ethylene or propylene oxide with fatty alcohols, phenols,
- Chelating agents beneficially aid cleaning and bleach stability by keeping metal ions, such as calcium, magnesium, and heavy metal cations in solution.
- suitable chelating agents include sodium tripolyphosphate, sodium acid pyrophosphate, tetrasodium pyrophosphate, aminopolycarboxylates such as nitrilotriacetic acid and ethylenediamine tetracetic acid and salts thereof, and polyphosphonates and aminopolyphosphonates such as hydroxyethanediphosphonic acid, ethylenediamine tetramethylenephosphonic acid, diethylenetriaminepentamethylenephosphonic acid and salts thereof.
- the chelating agent selected is not critical except that it must be compatible with the other ingredients ofthe denture cleanser when in the dry state and in aqueous solution.
- the chelating agent comprises between 0.1 and 60 percent by weight ofthe composition and preferably between 0.5 and 30 percent.
- Phosphonic acid chelating agents preferably comprise from about 0.1 to about 1 percent, preferably from about 0.1% to about 0.5% by weight of composition.
- Enzymes suitable for use herein are exemplified by proteases, alkalases, amylases, Upases, dextranases, mutanases, glucanases etc.
- the following are representative denture cleansing tablets according to the invention.
- the percentages are by weight of the total tablet.
- the tablets are made by compressing a mixture ofthe granulated components in a punch and dye tabletting press at a pressure of about 10-5 kPa.
- the overall tablet weight is 3 g; diameter 25 mm.
- the denture cleansing tablets of Examples I to V display improved antiplaque, cleansing and anti-bacterial activity together with excellent cohesion and other physical and in-use performance characteristics.
- the following are representative toothpaste/denture cleansing pastes according to the invention.
- the percentages are by weight of total composition.
- the toothpastes/denture cleansing pastes of Examples VI to VIII display improved antiplaque, flavour impact and anti-bacterial activity together with excellent cleansing characteristics.
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US08/981,949 US6193958B1 (en) | 1995-07-05 | 1996-06-25 | Oral compositions |
| JP9505174A JPH11509841A (en) | 1995-07-05 | 1996-06-25 | Oral composition |
| EP96921765A EP0836470A4 (en) | 1995-07-05 | 1996-06-25 | Oral compositions |
| CA002226160A CA2226160C (en) | 1995-07-05 | 1996-06-25 | Oral compositions |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB9513694.1 | 1995-07-05 | ||
| GBGB9513694.1A GB9513694D0 (en) | 1995-07-05 | 1995-07-05 | Oral compositions |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO1997002011A1 true WO1997002011A1 (en) | 1997-01-23 |
Family
ID=10777164
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US1996/010815 WO1997002011A1 (en) | 1995-07-05 | 1996-06-25 | Oral compositions |
Country Status (5)
| Country | Link |
|---|---|
| EP (1) | EP0836470A4 (en) |
| JP (1) | JPH11509841A (en) |
| CA (1) | CA2226160C (en) |
| GB (1) | GB9513694D0 (en) |
| WO (1) | WO1997002011A1 (en) |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2140332A1 (en) * | 1997-12-22 | 2000-02-16 | Compania Anonima De Importacio | Toothpaste in the form of chewing gum. |
| AU732560B2 (en) * | 1997-12-23 | 2001-04-26 | Wm. Wrigley Jr. Company | Chewing gum and gum bases with reduced adhesion to environmental surfaces |
| WO2001039606A1 (en) * | 1999-11-12 | 2001-06-07 | The Procter & Gamble Company | Chewing gum compositions |
| WO2001056399A1 (en) * | 2000-02-04 | 2001-08-09 | The Procter & Gamble Company | Chewing gum compositions |
| WO2001067884A1 (en) * | 2000-03-10 | 2001-09-20 | Warner-Lambert Company | Stain removing chewing gum and confectionery compositions, and methods of making and using the same |
| EP1215494A3 (en) * | 1997-04-30 | 2002-09-18 | Becton Dickinson and Company | Additive preparation for blood sample and method of use thereof |
| US6485739B2 (en) | 2000-03-10 | 2002-11-26 | Warner-Lambert Company | Stain removing chewing gum and confectionery compositions, and methods of making and using the same |
| US6670312B2 (en) | 2000-12-08 | 2003-12-30 | Takeshi Sugimoto | Composition for removal of calcium or magnesium compounds from an article |
| AU2002313994B2 (en) * | 1997-12-23 | 2005-05-05 | Wm. Wrigley Jr. Company | Chewing Gum and Gum Bases having Reduced Adhesion to Environmental Surfaces |
| AU2002300487B2 (en) * | 1997-04-30 | 2006-04-27 | Becton, Dickinson And Company | Additive preparation and method of use thereof |
| US7727565B2 (en) | 2004-08-25 | 2010-06-01 | Cadbury Adams Usa Llc | Liquid-filled chewing gum composition |
| US8133476B2 (en) | 2006-04-05 | 2012-03-13 | Cadbury Adams Usa Llc | Calcium phosphate complex and salts in oral delivery systems |
| US8252269B2 (en) | 2006-04-05 | 2012-08-28 | Cadbury Adams Usa, Llc | Impact of calcium phosphate complex on dental caries |
| US9011946B2 (en) | 2011-04-29 | 2015-04-21 | Intercontinental Great Brands Llc | Encapsulated acid, method for the preparation thereof, and chewing gum comprising same |
| US9198448B2 (en) | 2005-02-07 | 2015-12-01 | Intercontinental Great Brands Llc | Stable tooth whitening gum with reactive ingredients |
| US9271904B2 (en) | 2003-11-21 | 2016-03-01 | Intercontinental Great Brands Llc | Controlled release oral delivery systems |
Families Citing this family (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE367432T1 (en) * | 2001-11-09 | 2007-08-15 | Jiro Sakurai | DETERGENT FOR METALLIC PRODUCT |
| JP6088301B2 (en) * | 2013-03-12 | 2017-03-01 | 小林製薬株式会社 | Denture cleaning agent |
| JP6953707B2 (en) * | 2016-11-30 | 2021-10-27 | ライオン株式会社 | Liquid denture cleanser composition |
| JP6926457B2 (en) * | 2016-11-30 | 2021-08-25 | ライオン株式会社 | Concentrated liquid denture cleanser composition |
| KR20190089841A (en) * | 2016-11-30 | 2019-07-31 | 라이온 가부시키가이샤 | Liquid denture cleanser composition, denture cleansing set, and denture cleansing method |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4994593A (en) * | 1988-11-28 | 1991-02-19 | Chesebrough-Pond's Usa Co. Division Of Conopco, Inc. | Hydroxylhydrocarbyl-modified aminoalkyl silicones |
| US5078988A (en) * | 1988-11-28 | 1992-01-07 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Dentrifrices including modified aminoalkyl silicones |
| US5154915A (en) * | 1988-11-28 | 1992-10-13 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Dentifrices containing aminoalkyl silicones and sarcosinate surfactants |
| US5188822A (en) * | 1991-08-07 | 1993-02-23 | Chesebrough-Pond's Usa Co., Division Of Conopco Inc. | Oral compositions containing an aminosilicone and a lipophilic compound |
| US5427770A (en) * | 1988-11-28 | 1995-06-27 | Chesebrough-Ponds Usa Co., Division Of Conopco, Inc. | Dentifrices containing amino alkyl silicones |
-
1995
- 1995-07-05 GB GBGB9513694.1A patent/GB9513694D0/en active Pending
-
1996
- 1996-06-25 WO PCT/US1996/010815 patent/WO1997002011A1/en not_active Application Discontinuation
- 1996-06-25 JP JP9505174A patent/JPH11509841A/en active Pending
- 1996-06-25 EP EP96921765A patent/EP0836470A4/en not_active Withdrawn
- 1996-06-25 CA CA002226160A patent/CA2226160C/en not_active Expired - Lifetime
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4994593A (en) * | 1988-11-28 | 1991-02-19 | Chesebrough-Pond's Usa Co. Division Of Conopco, Inc. | Hydroxylhydrocarbyl-modified aminoalkyl silicones |
| US5078988A (en) * | 1988-11-28 | 1992-01-07 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Dentrifrices including modified aminoalkyl silicones |
| US5154915A (en) * | 1988-11-28 | 1992-10-13 | Chesebrough-Pond's Usa Co., Division Of Conopco, Inc. | Dentifrices containing aminoalkyl silicones and sarcosinate surfactants |
| US5427770A (en) * | 1988-11-28 | 1995-06-27 | Chesebrough-Ponds Usa Co., Division Of Conopco, Inc. | Dentifrices containing amino alkyl silicones |
| US5188822A (en) * | 1991-08-07 | 1993-02-23 | Chesebrough-Pond's Usa Co., Division Of Conopco Inc. | Oral compositions containing an aminosilicone and a lipophilic compound |
Non-Patent Citations (1)
| Title |
|---|
| See also references of EP0836470A4 * |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6534016B1 (en) | 1997-04-30 | 2003-03-18 | Richmond Cohen | Additive preparation and method of use thereof |
| AU2002300487B2 (en) * | 1997-04-30 | 2006-04-27 | Becton, Dickinson And Company | Additive preparation and method of use thereof |
| EP1215494A3 (en) * | 1997-04-30 | 2002-09-18 | Becton Dickinson and Company | Additive preparation for blood sample and method of use thereof |
| ES2140332A1 (en) * | 1997-12-22 | 2000-02-16 | Compania Anonima De Importacio | Toothpaste in the form of chewing gum. |
| AU732560B2 (en) * | 1997-12-23 | 2001-04-26 | Wm. Wrigley Jr. Company | Chewing gum and gum bases with reduced adhesion to environmental surfaces |
| AU2002313994B2 (en) * | 1997-12-23 | 2005-05-05 | Wm. Wrigley Jr. Company | Chewing Gum and Gum Bases having Reduced Adhesion to Environmental Surfaces |
| WO2001039606A1 (en) * | 1999-11-12 | 2001-06-07 | The Procter & Gamble Company | Chewing gum compositions |
| WO2001056399A1 (en) * | 2000-02-04 | 2001-08-09 | The Procter & Gamble Company | Chewing gum compositions |
| US6696044B2 (en) | 2000-03-10 | 2004-02-24 | Cadbury Adams Usa Llc | Stain removing chewing gum and confectionery compositions, and methods of making and using the same |
| US6485739B2 (en) | 2000-03-10 | 2002-11-26 | Warner-Lambert Company | Stain removing chewing gum and confectionery compositions, and methods of making and using the same |
| US6471945B2 (en) | 2000-03-10 | 2002-10-29 | Warner-Lambert Company | Stain removing chewing gum and confectionery compositions, and methods of making and using the same |
| WO2001067884A1 (en) * | 2000-03-10 | 2001-09-20 | Warner-Lambert Company | Stain removing chewing gum and confectionery compositions, and methods of making and using the same |
| US6670312B2 (en) | 2000-12-08 | 2003-12-30 | Takeshi Sugimoto | Composition for removal of calcium or magnesium compounds from an article |
| US9271904B2 (en) | 2003-11-21 | 2016-03-01 | Intercontinental Great Brands Llc | Controlled release oral delivery systems |
| US7727565B2 (en) | 2004-08-25 | 2010-06-01 | Cadbury Adams Usa Llc | Liquid-filled chewing gum composition |
| US9198448B2 (en) | 2005-02-07 | 2015-12-01 | Intercontinental Great Brands Llc | Stable tooth whitening gum with reactive ingredients |
| US8133476B2 (en) | 2006-04-05 | 2012-03-13 | Cadbury Adams Usa Llc | Calcium phosphate complex and salts in oral delivery systems |
| US8252269B2 (en) | 2006-04-05 | 2012-08-28 | Cadbury Adams Usa, Llc | Impact of calcium phosphate complex on dental caries |
| US9011946B2 (en) | 2011-04-29 | 2015-04-21 | Intercontinental Great Brands Llc | Encapsulated acid, method for the preparation thereof, and chewing gum comprising same |
| US9737082B2 (en) | 2011-04-29 | 2017-08-22 | Intercontinental Great Brands Llc | Chewing gum composition comprising encapsulated acid |
Also Published As
| Publication number | Publication date |
|---|---|
| GB9513694D0 (en) | 1995-09-06 |
| CA2226160C (en) | 2002-04-30 |
| CA2226160A1 (en) | 1997-01-23 |
| JPH11509841A (en) | 1999-08-31 |
| EP0836470A4 (en) | 1999-10-13 |
| EP0836470A1 (en) | 1998-04-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US5827505A (en) | Oral compositions | |
| US6004538A (en) | Oral compositions | |
| EP0863742B1 (en) | Granular powders containing silicone oil | |
| CA2226160C (en) | Oral compositions | |
| AU4517496A (en) | Oral compositions | |
| CA2208364C (en) | Oral compositions | |
| US6193958B1 (en) | Oral compositions | |
| EP0801555B1 (en) | Oral compositions containing dimethicone copolyols | |
| US6294154B1 (en) | Oral compositions containing dimethicone copolyols | |
| CA2216726C (en) | Oral compositions | |
| AU4470196A (en) | Oral compositions | |
| AU727373B2 (en) | Oral compositions | |
| AU4476499A (en) | Oral compositions | |
| MXPA97004663A (en) | Ora compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A1 Designated state(s): CA CN JP NO TR US |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A1 Designated state(s): AT BE CH DE DK ES FI FR GB GR IE IT LU MC NL PT SE |
|
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| ENP | Entry into the national phase |
Ref document number: 2226160 Country of ref document: CA Ref country code: CA Ref document number: 2226160 Kind code of ref document: A Format of ref document f/p: F |
|
| ENP | Entry into the national phase |
Ref country code: JP Ref document number: 1997 505174 Kind code of ref document: A Format of ref document f/p: F |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1996921765 Country of ref document: EP |
|
| WWP | Wipo information: published in national office |
Ref document number: 1996921765 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08981949 Country of ref document: US |
|
| WWW | Wipo information: withdrawn in national office |
Ref document number: 1996921765 Country of ref document: EP |