WO2001025187A2 - Benzylamidoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide - Google Patents
Benzylamidoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide Download PDFInfo
- Publication number
- WO2001025187A2 WO2001025187A2 PCT/EP2000/009744 EP0009744W WO0125187A2 WO 2001025187 A2 WO2001025187 A2 WO 2001025187A2 EP 0009744 W EP0009744 W EP 0009744W WO 0125187 A2 WO0125187 A2 WO 0125187A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- formula
- group
- alkoxy
- halogen
- Prior art date
Links
- -1 Benzyl amidoxime derivatives Chemical class 0.000 title claims abstract description 68
- 239000000417 fungicide Substances 0.000 title claims abstract description 12
- 238000000034 method Methods 0.000 title claims description 6
- 239000013067 intermediate product Substances 0.000 title description 2
- 229910052736 halogen Inorganic materials 0.000 claims abstract description 28
- 150000002367 halogens Chemical class 0.000 claims abstract description 28
- 239000001257 hydrogen Substances 0.000 claims abstract description 17
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract description 17
- 229910052799 carbon Inorganic materials 0.000 claims abstract description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract description 13
- 229910052757 nitrogen Inorganic materials 0.000 claims abstract description 10
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims abstract description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims abstract description 8
- 125000003118 aryl group Chemical group 0.000 claims abstract description 6
- 229910052760 oxygen Inorganic materials 0.000 claims abstract description 6
- 239000001301 oxygen Substances 0.000 claims abstract description 6
- 125000005083 alkoxyalkoxy group Chemical group 0.000 claims abstract description 5
- 125000004432 carbon atom Chemical group C* 0.000 claims abstract description 5
- 125000004438 haloalkoxy group Chemical group 0.000 claims abstract description 5
- 125000001188 haloalkyl group Chemical group 0.000 claims abstract description 5
- 150000001875 compounds Chemical class 0.000 claims description 30
- 125000001424 substituent group Chemical group 0.000 claims description 17
- 238000002360 preparation method Methods 0.000 claims description 11
- 150000002431 hydrogen Chemical class 0.000 claims description 10
- 125000001544 thienyl group Chemical group 0.000 claims description 10
- 125000000217 alkyl group Chemical group 0.000 claims description 9
- 241000233866 Fungi Species 0.000 claims description 8
- 229910052717 sulfur Inorganic materials 0.000 claims description 8
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 6
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 6
- 150000003839 salts Chemical class 0.000 claims description 5
- 125000004765 (C1-C4) haloalkyl group Chemical group 0.000 claims description 4
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 claims description 4
- 239000000463 material Substances 0.000 claims description 4
- 239000002253 acid Substances 0.000 claims description 3
- 125000004076 pyridyl group Chemical group 0.000 claims description 3
- 125000004434 sulfur atom Chemical group 0.000 claims description 3
- 239000007864 aqueous solution Substances 0.000 claims description 2
- SFZULDYEOVSIKM-UHFFFAOYSA-N chembl321317 Chemical class C1=CC(C(=N)NO)=CC=C1C1=CC=C(C=2C=CC(=CC=2)C(=N)NO)O1 SFZULDYEOVSIKM-UHFFFAOYSA-N 0.000 claims description 2
- MXOQNVMDKHLYCZ-UHFFFAOYSA-N benzamidoxime Chemical class ON=C(N)C1=CC=CC=C1 MXOQNVMDKHLYCZ-UHFFFAOYSA-N 0.000 claims 8
- 239000000654 additive Substances 0.000 claims 1
- 239000012872 agrochemical composition Substances 0.000 claims 1
- 150000008359 benzonitriles Chemical class 0.000 claims 1
- 150000004820 halides Chemical class 0.000 claims 1
- 125000005843 halogen group Chemical group 0.000 claims 1
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical group [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 abstract description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 abstract description 2
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 abstract 2
- 125000004414 alkyl thio group Chemical group 0.000 abstract 2
- 125000004435 hydrogen atom Chemical class [H]* 0.000 abstract 2
- 125000004916 (C1-C6) alkylcarbonyl group Chemical group 0.000 abstract 1
- 125000002947 alkylene group Chemical group 0.000 abstract 1
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 15
- 239000004480 active ingredient Substances 0.000 description 15
- 241000196324 Embryophyta Species 0.000 description 13
- 239000000203 mixture Substances 0.000 description 13
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 13
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 12
- 150000001721 carbon Chemical group 0.000 description 9
- 239000000460 chlorine Substances 0.000 description 9
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 8
- 229910052801 chlorine Inorganic materials 0.000 description 8
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- 239000011737 fluorine Substances 0.000 description 7
- 229910052731 fluorine Inorganic materials 0.000 description 7
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 7
- 150000003254 radicals Chemical class 0.000 description 7
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N EtOH Substances CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 6
- 239000000284 extract Substances 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 5
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 5
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 5
- 239000006185 dispersion Substances 0.000 description 5
- 239000000741 silica gel Substances 0.000 description 5
- 229910002027 silica gel Inorganic materials 0.000 description 5
- 229910052938 sodium sulfate Inorganic materials 0.000 description 5
- 235000011152 sodium sulphate Nutrition 0.000 description 5
- 235000013311 vegetables Nutrition 0.000 description 5
- QGZKDVFQNNGYKY-UHFFFAOYSA-N Ammonia Chemical compound N QGZKDVFQNNGYKY-UHFFFAOYSA-N 0.000 description 4
- 240000008067 Cucumis sativus Species 0.000 description 4
- 241000221785 Erysiphales Species 0.000 description 4
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 4
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 4
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 4
- 241000209140 Triticum Species 0.000 description 4
- 235000021307 Triticum Nutrition 0.000 description 4
- 230000000694 effects Effects 0.000 description 4
- 230000000855 fungicidal effect Effects 0.000 description 4
- 125000005842 heteroatom Chemical group 0.000 description 4
- 229910000403 monosodium phosphate Inorganic materials 0.000 description 4
- 235000019799 monosodium phosphate Nutrition 0.000 description 4
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 4
- 159000000000 sodium salts Chemical class 0.000 description 4
- 241000894007 species Species 0.000 description 4
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 3
- 235000010799 Cucumis sativus var sativus Nutrition 0.000 description 3
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 3
- 206010061217 Infestation Diseases 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 229910052794 bromium Inorganic materials 0.000 description 3
- AEILLAXRDHDKDY-UHFFFAOYSA-N bromomethylcyclopropane Chemical compound BrCC1CC1 AEILLAXRDHDKDY-UHFFFAOYSA-N 0.000 description 3
- 235000013339 cereals Nutrition 0.000 description 3
- 239000000543 intermediate Substances 0.000 description 3
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 3
- AEXITZJSLGALNH-UHFFFAOYSA-N n'-hydroxyethanimidamide Chemical compound CC(N)=NO AEXITZJSLGALNH-UHFFFAOYSA-N 0.000 description 3
- 239000008363 phosphate buffer Substances 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000002689 soil Substances 0.000 description 3
- 239000007787 solid Substances 0.000 description 3
- 239000011593 sulfur Substances 0.000 description 3
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 3
- 125000004201 2,4-dichlorophenyl group Chemical group [H]C1=C([H])C(*)=C(Cl)C([H])=C1Cl 0.000 description 2
- WBIQQQGBSDOWNP-UHFFFAOYSA-N 2-dodecylbenzenesulfonic acid Chemical compound CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O WBIQQQGBSDOWNP-UHFFFAOYSA-N 0.000 description 2
- VMZCDNSFRSVYKQ-UHFFFAOYSA-N 2-phenylacetyl chloride Chemical compound ClC(=O)CC1=CC=CC=C1 VMZCDNSFRSVYKQ-UHFFFAOYSA-N 0.000 description 2
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 2
- DLFVBJFMPXGRIB-UHFFFAOYSA-N Acetamide Chemical compound CC(N)=O DLFVBJFMPXGRIB-UHFFFAOYSA-N 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 2
- 241000235349 Ascomycota Species 0.000 description 2
- 241000221198 Basidiomycota Species 0.000 description 2
- 229920000742 Cotton Polymers 0.000 description 2
- 240000004244 Cucurbita moschata Species 0.000 description 2
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 2
- 241000221787 Erysiphe Species 0.000 description 2
- 244000299507 Gossypium hirsutum Species 0.000 description 2
- 240000005979 Hordeum vulgare Species 0.000 description 2
- 235000007340 Hordeum vulgare Nutrition 0.000 description 2
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 2
- 244000070406 Malus silvestris Species 0.000 description 2
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 2
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 2
- 240000007594 Oryza sativa Species 0.000 description 2
- 235000007164 Oryza sativa Nutrition 0.000 description 2
- ZLMJMSJWJFRBEC-UHFFFAOYSA-N Potassium Chemical compound [K] ZLMJMSJWJFRBEC-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 241000579741 Sphaerotheca <fungi> Species 0.000 description 2
- 229920001807 Urea-formaldehyde Polymers 0.000 description 2
- 229910021529 ammonia Inorganic materials 0.000 description 2
- 235000021016 apples Nutrition 0.000 description 2
- UHOVQNZJYSORNB-UHFFFAOYSA-N benzene Substances C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 238000009835 boiling Methods 0.000 description 2
- 159000000007 calcium salts Chemical class 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 238000004587 chromatography analysis Methods 0.000 description 2
- 239000012043 crude product Substances 0.000 description 2
- 125000004786 difluoromethoxy group Chemical group [H]C(F)(F)O* 0.000 description 2
- 239000012990 dithiocarbamate Substances 0.000 description 2
- 229940060296 dodecylbenzenesulfonic acid Drugs 0.000 description 2
- 235000013399 edible fruits Nutrition 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 239000003337 fertilizer Substances 0.000 description 2
- 235000013312 flour Nutrition 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 125000001072 heteroaryl group Chemical group 0.000 description 2
- 239000012442 inert solvent Substances 0.000 description 2
- 208000015181 infectious disease Diseases 0.000 description 2
- 239000011872 intimate mixture Substances 0.000 description 2
- ZXEKIIBDNHEJCQ-UHFFFAOYSA-N isobutanol Chemical compound CC(C)CO ZXEKIIBDNHEJCQ-UHFFFAOYSA-N 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 235000010446 mineral oil Nutrition 0.000 description 2
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 239000003921 oil Substances 0.000 description 2
- 230000003032 phytopathogenic effect Effects 0.000 description 2
- 239000011591 potassium Substances 0.000 description 2
- 229910052700 potassium Inorganic materials 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 235000009566 rice Nutrition 0.000 description 2
- 235000017557 sodium bicarbonate Nutrition 0.000 description 2
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- 239000002904 solvent Substances 0.000 description 2
- 238000005507 spraying Methods 0.000 description 2
- 239000011550 stock solution Substances 0.000 description 2
- 239000002023 wood Substances 0.000 description 2
- 239000008096 xylene Substances 0.000 description 2
- 125000004767 (C1-C4) haloalkoxy group Chemical group 0.000 description 1
- UTDRFZYLWMKWGN-UHFFFAOYSA-N 1,3-bis(2-methylpropyl)naphthalene-2-sulfonic acid Chemical compound C1=CC=C2C(CC(C)C)=C(S(O)(=O)=O)C(CC(C)C)=CC2=C1 UTDRFZYLWMKWGN-UHFFFAOYSA-N 0.000 description 1
- HNEGJTWNOOWEMH-UHFFFAOYSA-N 1-fluoropropane Chemical group [CH2]CCF HNEGJTWNOOWEMH-UHFFFAOYSA-N 0.000 description 1
- 125000004343 1-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- 125000004778 2,2-difluoroethyl group Chemical group [H]C([H])(*)C([H])(F)F 0.000 description 1
- SNTWKPAKVQFCCF-UHFFFAOYSA-N 2,3-dihydro-1h-triazole Chemical compound N1NC=CN1 SNTWKPAKVQFCCF-UHFFFAOYSA-N 0.000 description 1
- IUWQJWIKCTXFIT-PJQLUOCWSA-N 2,3-dinitrooctan-2-yl (e)-2-phenylbut-2-enoate Chemical compound CCCCCC([N+]([O-])=O)C(C)([N+]([O-])=O)OC(=O)C(=C\C)\C1=CC=CC=C1 IUWQJWIKCTXFIT-PJQLUOCWSA-N 0.000 description 1
- NZUXRGMXFCTGBV-UHFFFAOYSA-N 2-(1,1,2,2-tetrachloroethylsulfanyl)-3a,4,5,7a-tetrahydroisoindole-1,3-dione Chemical compound C1CC=CC2C(=O)N(SC(Cl)(Cl)C(Cl)Cl)C(=O)C21 NZUXRGMXFCTGBV-UHFFFAOYSA-N 0.000 description 1
- CMBQTCSOPKKWEX-UHFFFAOYSA-N 2-(2-chloro-6-fluorophenyl)-n'-hydroxyethanimidamide Chemical compound ON=C(N)CC1=C(F)C=CC=C1Cl CMBQTCSOPKKWEX-UHFFFAOYSA-N 0.000 description 1
- PAWQVTBBRAZDMG-UHFFFAOYSA-N 2-(3-bromo-2-fluorophenyl)acetic acid Chemical compound OC(=O)CC1=CC=CC(Br)=C1F PAWQVTBBRAZDMG-UHFFFAOYSA-N 0.000 description 1
- AILMSPBSXLNSLU-UHFFFAOYSA-N 2-(4-chlorophenyl)-3-(3,4-dimethoxyphenyl)-1-morpholin-4-ylprop-2-en-1-one Chemical compound C1=C(OC)C(OC)=CC=C1C=C(C=1C=CC(Cl)=CC=1)C(=O)N1CCOCC1 AILMSPBSXLNSLU-UHFFFAOYSA-N 0.000 description 1
- IQHSSYROJYPFDV-UHFFFAOYSA-N 2-bromo-1,3-dichloro-5-(trifluoromethyl)benzene Chemical group FC(F)(F)C1=CC(Cl)=C(Br)C(Cl)=C1 IQHSSYROJYPFDV-UHFFFAOYSA-N 0.000 description 1
- 125000005999 2-bromoethyl group Chemical group 0.000 description 1
- 125000004780 2-chloro-2,2-difluoroethyl group Chemical group [H]C([H])(*)C(F)(F)Cl 0.000 description 1
- 125000004779 2-chloro-2-fluoroethyl group Chemical group [H]C([H])(*)C([H])(F)Cl 0.000 description 1
- 125000001340 2-chloroethyl group Chemical group [H]C([H])(Cl)C([H])([H])* 0.000 description 1
- KKZUMAMOMRDVKA-UHFFFAOYSA-N 2-chloropropane Chemical group [CH2]C(C)Cl KKZUMAMOMRDVKA-UHFFFAOYSA-N 0.000 description 1
- 125000004777 2-fluoroethyl group Chemical group [H]C([H])(F)C([H])([H])* 0.000 description 1
- VLRGXXKFHVJQOL-UHFFFAOYSA-N 3-chloropentane-2,4-dione Chemical compound CC(=O)C(Cl)C(C)=O VLRGXXKFHVJQOL-UHFFFAOYSA-N 0.000 description 1
- CDIJOYCNNFLOAX-UHFFFAOYSA-N 4-(trichloromethylsulfanyl)isoindole-1,3-dione Chemical compound ClC(Cl)(Cl)SC1=CC=CC2=C1C(=O)NC2=O CDIJOYCNNFLOAX-UHFFFAOYSA-N 0.000 description 1
- OOTHTARUZHONSW-UHFFFAOYSA-N 4-[(2-chlorophenyl)hydrazinylidene]-3-methyl-1,2-oxazol-5-one Chemical compound CC1=NOC(=O)C1=NNC1=CC=CC=C1Cl OOTHTARUZHONSW-UHFFFAOYSA-N 0.000 description 1
- 125000004070 6 membered heterocyclic group Chemical group 0.000 description 1
- 241000223600 Alternaria Species 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 239000004254 Ammonium phosphate Substances 0.000 description 1
- 244000105624 Arachis hypogaea Species 0.000 description 1
- 235000007319 Avena orientalis Nutrition 0.000 description 1
- 244000075850 Avena orientalis Species 0.000 description 1
- 235000000832 Ayote Nutrition 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-M Bisulfite Chemical compound OS([O-])=O LSNNMFCWUKXFEE-UHFFFAOYSA-M 0.000 description 1
- 241000123650 Botrytis cinerea Species 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical group [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 240000007154 Coffea arabica Species 0.000 description 1
- 235000009849 Cucumis sativus Nutrition 0.000 description 1
- 235000009854 Cucurbita moschata Nutrition 0.000 description 1
- 235000009804 Cucurbita pepo subsp pepo Nutrition 0.000 description 1
- 240000009088 Fragaria x ananassa Species 0.000 description 1
- QTDRLOKFLJJHTG-UHFFFAOYSA-N Furmecyclox Chemical compound C1=C(C)OC(C)=C1C(=O)N(OC)C1CCCCC1 QTDRLOKFLJJHTG-UHFFFAOYSA-N 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 241000896246 Golovinomyces cichoracearum Species 0.000 description 1
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical compound ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 1
- 235000007688 Lycopersicon esculentum Nutrition 0.000 description 1
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 1
- MZNCVTCEYXDDIS-UHFFFAOYSA-N Mebenil Chemical compound CC1=CC=CC=C1C(=O)NC1=CC=CC=C1 MZNCVTCEYXDDIS-UHFFFAOYSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- 241000498271 Necator Species 0.000 description 1
- 241001668536 Oculimacula yallundae Species 0.000 description 1
- 241001236817 Paecilomyces <Clavicipitaceae> Species 0.000 description 1
- 241000315044 Passalora arachidicola Species 0.000 description 1
- 244000046052 Phaseolus vulgaris Species 0.000 description 1
- 235000010627 Phaseolus vulgaris Nutrition 0.000 description 1
- 241000233614 Phytophthora Species 0.000 description 1
- 241001281803 Plasmopara viticola Species 0.000 description 1
- 241000896242 Podosphaera Species 0.000 description 1
- 208000021068 Pulmonary arterial hypertension associated with portal hypertension Diseases 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- 241001361634 Rhizoctonia Species 0.000 description 1
- 240000000111 Saccharum officinarum Species 0.000 description 1
- 235000007201 Saccharum officinarum Nutrition 0.000 description 1
- 206010039509 Scab Diseases 0.000 description 1
- 244000082988 Secale cereale Species 0.000 description 1
- 235000007238 Secale cereale Nutrition 0.000 description 1
- 241000270295 Serpentes Species 0.000 description 1
- 240000003768 Solanum lycopersicum Species 0.000 description 1
- 244000061456 Solanum tuberosum Species 0.000 description 1
- 235000002595 Solanum tuberosum Nutrition 0.000 description 1
- LSNNMFCWUKXFEE-UHFFFAOYSA-N Sulfurous acid Chemical compound OS(O)=O LSNNMFCWUKXFEE-UHFFFAOYSA-N 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric Acid Chemical compound [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 240000008042 Zea mays Species 0.000 description 1
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 description 1
- 235000002017 Zea mays subsp mays Nutrition 0.000 description 1
- HCHKCACWOHOZIP-UHFFFAOYSA-N Zinc Chemical compound [Zn] HCHKCACWOHOZIP-UHFFFAOYSA-N 0.000 description 1
- 150000008065 acid anhydrides Chemical class 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 125000003545 alkoxy group Chemical group 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 229910000148 ammonium phosphate Inorganic materials 0.000 description 1
- 235000019289 ammonium phosphates Nutrition 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 1
- 244000052616 bacterial pathogen Species 0.000 description 1
- LJOZMWRYMKECFF-UHFFFAOYSA-N benodanil Chemical compound IC1=CC=CC=C1C(=O)NC1=CC=CC=C1 LJOZMWRYMKECFF-UHFFFAOYSA-N 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000004202 carbamide Substances 0.000 description 1
- 235000013877 carbamide Nutrition 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 150000001805 chlorine compounds Chemical class 0.000 description 1
- 125000004773 chlorofluoromethyl group Chemical group [H]C(F)(Cl)* 0.000 description 1
- 125000004218 chloromethyl group Chemical group [H]C([H])(Cl)* 0.000 description 1
- ZVTQWXCKQTUVPY-UHFFFAOYSA-N chloromethylcyclopropane Chemical compound ClCC1CC1 ZVTQWXCKQTUVPY-UHFFFAOYSA-N 0.000 description 1
- APEJMQOBVMLION-UHFFFAOYSA-N cinnamamide Chemical class NC(=O)C=CC1=CC=CC=C1 APEJMQOBVMLION-UHFFFAOYSA-N 0.000 description 1
- 235000016213 coffee Nutrition 0.000 description 1
- 235000013353 coffee beverage Nutrition 0.000 description 1
- 239000013256 coordination polymer Substances 0.000 description 1
- 150000001879 copper Chemical class 0.000 description 1
- 235000005822 corn Nutrition 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- 125000004186 cyclopropylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C1([H])[H] 0.000 description 1
- MNNHAPBLZZVQHP-UHFFFAOYSA-N diammonium hydrogen phosphate Chemical compound [NH4+].[NH4+].OP([O-])([O-])=O MNNHAPBLZZVQHP-UHFFFAOYSA-N 0.000 description 1
- 125000004774 dichlorofluoromethyl group Chemical group FC(Cl)(Cl)* 0.000 description 1
- 125000004772 dichloromethyl group Chemical group [H]C(Cl)(Cl)* 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 150000004659 dithiocarbamates Chemical class 0.000 description 1
- 239000000428 dust Substances 0.000 description 1
- 238000010410 dusting Methods 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 150000002191 fatty alcohols Chemical class 0.000 description 1
- MUJOIMFVNIBMKC-UHFFFAOYSA-N fludioxonil Chemical compound C=12OC(F)(F)OC2=CC=CC=1C1=CNC=C1C#N MUJOIMFVNIBMKC-UHFFFAOYSA-N 0.000 description 1
- 125000004785 fluoromethoxy group Chemical group [H]C([H])(F)O* 0.000 description 1
- 125000004216 fluoromethyl group Chemical group [H]C([H])(F)* 0.000 description 1
- ZHNUHDYFZUAESO-UHFFFAOYSA-N formamide Substances NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 1
- 230000002538 fungal effect Effects 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 239000003630 growth substance Substances 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000006343 heptafluoro propyl group Chemical group 0.000 description 1
- 239000004009 herbicide Substances 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- CKAPSXZOOQJIBF-UHFFFAOYSA-N hexachlorobenzene Chemical compound ClC1=C(Cl)C(Cl)=C(Cl)C(Cl)=C1Cl CKAPSXZOOQJIBF-UHFFFAOYSA-N 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 125000000879 imine group Chemical group 0.000 description 1
- 125000001841 imino group Chemical group [H]N=* 0.000 description 1
- 239000002917 insecticide Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 1
- 235000019341 magnesium sulphate Nutrition 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- YKSNLCVSTHTHJA-UHFFFAOYSA-L maneb Chemical compound [Mn+2].[S-]C(=S)NCCNC([S-])=S YKSNLCVSTHTHJA-UHFFFAOYSA-L 0.000 description 1
- 229910052748 manganese Inorganic materials 0.000 description 1
- 239000011572 manganese Substances 0.000 description 1
- OKKJLVBELUTLKV-UHFFFAOYSA-N methanol Substances OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- NMMNBCCKCZCSNU-UHFFFAOYSA-N n'-(2,6-dichlorophenyl)-n-hydroxyethanimidamide Chemical compound ONC(C)=NC1=C(Cl)C=CC=C1Cl NMMNBCCKCZCSNU-UHFFFAOYSA-N 0.000 description 1
- OZFLVRMSUOBHBW-UHFFFAOYSA-N n'-(2-chloro-6-fluorophenyl)-n-hydroxyethanimidamide Chemical compound ONC(C)=NC1=C(F)C=CC=C1Cl OZFLVRMSUOBHBW-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 125000005246 nonafluorobutyl group Chemical group FC(F)(F)C(F)(F)C(F)(F)C(F)(F)* 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid group Chemical group C(CCCCCCC\C=C/CCCCCCCC)(=O)O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 description 1
- 235000020232 peanut Nutrition 0.000 description 1
- 125000006340 pentafluoro ethyl group Chemical group FC(F)(F)C(F)(F)* 0.000 description 1
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 1
- 150000008060 phenylpyrroles Chemical class 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920000151 polyglycol Polymers 0.000 description 1
- 239000010695 polyglycol Substances 0.000 description 1
- 235000012015 potatoes Nutrition 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 235000015136 pumpkin Nutrition 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 235000021012 strawberries Nutrition 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 125000000542 sulfonic acid group Chemical group 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000003866 trichloromethyl group Chemical group ClC(Cl)(Cl)* 0.000 description 1
- 150000003672 ureas Chemical class 0.000 description 1
- 239000002699 waste material Substances 0.000 description 1
- 239000011701 zinc Substances 0.000 description 1
- 229910052725 zinc Inorganic materials 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C259/00—Compounds containing carboxyl groups, an oxygen atom of a carboxyl group being replaced by a nitrogen atom, this nitrogen atom being further bound to an oxygen atom and not being part of nitro or nitroso groups
- C07C259/12—Compounds containing carboxyl groups, an oxygen atom of a carboxyl group being replaced by a nitrogen atom, this nitrogen atom being further bound to an oxygen atom and not being part of nitro or nitroso groups with replacement of the other oxygen atom of the carboxyl group by nitrogen atoms, e.g. N-hydroxyamidines
- C07C259/14—Compounds containing carboxyl groups, an oxygen atom of a carboxyl group being replaced by a nitrogen atom, this nitrogen atom being further bound to an oxygen atom and not being part of nitro or nitroso groups with replacement of the other oxygen atom of the carboxyl group by nitrogen atoms, e.g. N-hydroxyamidines having carbon atoms of hydroxamidine groups bound to hydrogen atoms or to acyclic carbon atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/52—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing groups, e.g. carboxylic acid amidines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2601/00—Systems containing only non-condensed rings
- C07C2601/02—Systems containing only non-condensed rings with a three-membered ring
Definitions
- the present invention relates to new benzylamide oxime derivatives, processes and intermediates for their preparation and their use as fungicides.
- Japanese Patent Application Laid-Open JP 10-95771 describes, among other things, fungicidal benzylamide oximes which, however, cannot be completely satisfactory in terms of their fungicidal activity and biological properties.
- the object of the present invention was therefore to provide new Benza idoxim derivatives with improved biological properties and increased activity, in particular even at low application rates.
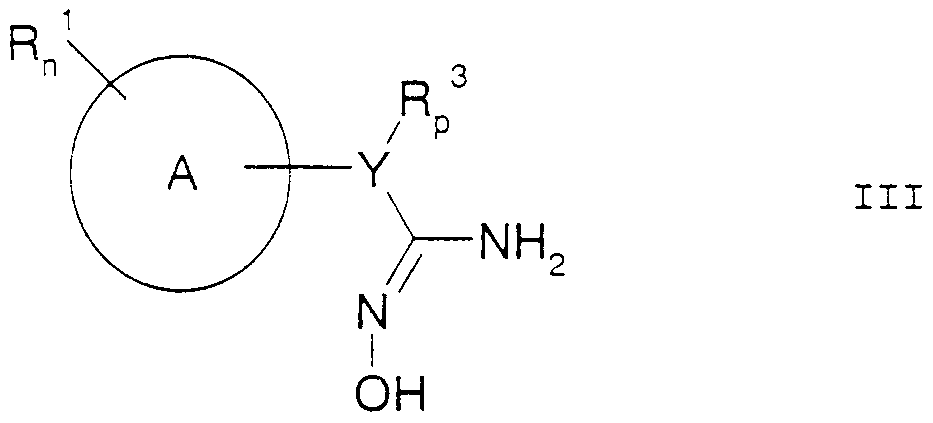
- the present application therefore relates to benzalamidoxime derivatives of the formula I.
- A is an aryl or hetaryl radical from the group phenyl, py ⁇ dyl or thienyl;
- R n 1 one to five identical or mixed residues from the group: hydrogen, halogen, Ci-Cg-alkyl, Ci-C ö alkoxy, C ⁇ -C -haloalkyl, C ⁇ -C -haloalkoxy, C ⁇ -C-alkylthio, C ⁇ -C alkoxyalkoxy;
- R 2 phenyl-C_-C ⁇ -alkyl which has one or more substituents selected from the group consisting of halogen, C ⁇ -C -alkyl, C ⁇ -C -haloalkyl, C -C 4 -alkoxy or C ⁇ -C 4 - Halogenalkoxy can wear, or
- Th ⁇ enyl-C ⁇ -C 4 -alkyl which on thienyl ring one or more substituents selected from the group consisting of halogen, C ⁇ -C -alkyl, C 4 haloalkyl, C ⁇ -C 4 alkoxy or C ⁇ -C 4 -haloalkoxy can wear or
- Pyrazolyl-C ⁇ -C 4 -alkyl which on the pyrazole ring one or more substituents selected from the group consisting of halogen, C ⁇ -C -alkyl, C haloalkyl, C ⁇ -C 4 alkoxy or C ⁇ -C -haloalkoxy carry 4 can
- R p 3 one to five identical or different radicals from the group: hydrogen, halogen, Ci-Cg-alkyl, Ci-C ß -alkoxy, C ⁇ -C 4 -haloalkyl, C ⁇ -C 4 -haiogenalkoxy, C ⁇ -C 4 - Alkylth ⁇ o, C ⁇ -C 4 alkoxyalkoxy, Ci-Cg-alkylcarbonyl;
- n 0, 1, 2, 3, 4 or 5;
- halogen is in each case fluorine, bromine, chlorine or iodine, in particular fluorine or chlorine.
- Ci-Cg-alkyl for: methyl, ethyl, n-propyl, 1-methylethyl, n-butyl, 1-methylpropyl, 2-methylpropyl or 1, 1-dimethylethyl, in particular for C ⁇ -C-alkyl, and for methyl or ethyl;
- ⁇ er is partially or completely substituted by fluorine, chlorine, bromine and / or iodine, ie for example chloromethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, dichlorofluoromethyl, chlorine difluoromethyl, 2-fluoroethyl, 2-chloroethyl, 2-bromoethyl, 2-iodoethyl, 2, 2-difluoroethyl, 2, 2, 2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro-2, 2-difluoroethyl, 2, 2-D ⁇ chlor-2-fluoroethyl, 2, 2, 2-Tr ⁇ chlorethyl, pentafluoroethyl, 2-fluoropropyl, 3-fluoropropyl,
- -C-C 4 alkylene for: a straight-chain or branched carbon chain, such as -CH 2 -, -CH 2 -CH 2 -, -CH (CH 3 ) -, -CH 2 -CH (CH 3 ) -, CH ( CH 3 ) -CH 2 -, CH 2 -CH 2 -CH 2 -, -CH 2 -CH (CH 3 ) -CH 2 -;
- C 1 -C 4 alkylene where a carbon atom can be replaced by an oxygen, sulfur or nitrogen atom, for: a C 1 -C 4 alkylene as mentioned above, where any carbon atom is replaced by a hetero atom
- X 0, S , NH
- X 0, S , NH
- Ci-Cg-alkoxy for: methoxy, ethoxy, n-propoxy, 1-methylethoxy, n-butoxy, 1-methylpropoxy, 2-methylpropoxy or 1, 1-dimethyl-ethoxy, in particular for -C-C 4 alkoxy, and for Methoxy or ethoxy;
- Ci-Cg-haloalkoxy for: a -CC 4 alkoxy radical as mentioned above, which is partially or completely substituted by fluorine, chlorine, bromine and / or iodine, so for example chloromethoxy, dichloromethoxy, T ⁇ chlormethoxy, fluoromethoxy, difluoromethoxy, trifluoromethoxy , Chlorofluoromethoxy, dichlorofluoromethoxy, chlorodifluoromethoxy, 2-fluoroethoxy, 2-chloroethoxy, 2-bromoethoxy, 2-iodoethoxy, 2, 2-difluoroethoxy, 2, 2, 2-tri
- Phenyl-C 1 -C 6 -alkyl for: for example benzyl, 1-phenylethyl, 2-phenylethyl, 1-phenylprop-l-yl, 2-phenylprop-l-yl, 3-phenylprop-1-yl, 1 - (Phenylethyl) -eth-l-yl, l- (phenylmethyl) -1- (methyl) -eth-l-yl or 1- (phenylmethyl) prop-1-yl, especially for benzyl or 2- Phenylethyl;
- Th ⁇ enyl-C 1 -C 4 alkyl for: for example 2-th ⁇ enylmethyl, 3-th ⁇ enylmethyl, 2-th ⁇ enylethyl, 2-th ⁇ enylprop-l-yl or 3-th ⁇ enylprop-1-yl;
- Pyrazole -CC 4 alkyl for: for example 1-pyrazolylmethyl, 2-pyrazolylmethyl, 3-pyrazolylmethyl, 2-pyrazolylylethyl, 2-pyrazolylprop-1-yl or 3-pyrazolylprop-l-yl;
- Heteroaryl an aromatic 5- or 6-membered heterocyclic ring which has one to four identical or different heteroatoms, selected from the following group: oxygen,
- Aryl an aromatic carbocyclic, mono- or bicyclic ring with 6-14 carbon atoms, such as phenyl, naphthyl; especially phenyl.
- R 1 here preferably denotes fluorine, chlorine, methyl, methoxy or trifluoromethyl.
- suostituents R 1 n 2,6-dichloro; 2-chloro-6-fluorine; 2,6-D ⁇ fluor; 2-chloro-5, 6-d ⁇ fluor; 2-chloro-6-t ⁇ fluoromethyl; 2-fluoro-6-t ⁇ fluoromethyl; 2-bromo-6-tn-fluoromethyl; 2-iodo-6-t ⁇ fluormethyl; 2,6-D ⁇ brom; 2-bromo-6-fluorine; 2-bromo-6-chloro; 2-chloro-6-trifluoromethoxy; 2-fluoro-6-trifluoromethoxy; 2-chloro-6-d ⁇ fluormethoxy; 2-D ⁇ fluormethoxy-6-fluoro; 2, 3-D ⁇ chlor-6-d ⁇ fluormethoxy; 2, 3-difluoro-6-difluoromethoxy; 2,6-B ⁇ s (difluoromethoxy);
- the group R 2 is preferably phenylmethyl; (4-chlorophenylmethyl; (4-fluorophenyl) methyl; (4-methylphenyl) methyl; (3-methylphenyl) ethyl; (4-trifluoromethylphenyl) methyl; (4-methoxyphenyl) methyl; 2-thenyl) methyl ,
- Y means in particular a straight-chain or branched C 1 -C 3 -alkylene chain, it being possible for a carbon atom to be replaced by an oxygen or sulfur atom or an imino group (-NH-) or alkyl imine group (-N (alkyl) -).
- A is an aryl or hetaryl group from the group phenyl, pyridyl or thienyl;
- Y is a carbon atom
- R n l one to five identical or different radicals from the group: hydrogen, halogen, C 1 -C 6 -alkyl, C 1 -C 6 alkoxy,
- R 2 is phenyl-C ⁇ -CG-alkyl, which Phenyl ⁇ ng one or more substituents C ⁇ -C selected from the group consisting of halogen, 4 alkyl, C 1 -C 4 haloalkyl, C ⁇ -C 4 alkoxy or C ⁇ - C 4 -haloalkoxy can wear, or
- Th ⁇ enyl-C ⁇ -C 4 alkyl which comprises one or more substituents on the thienyl ring, selected from the group consisting of halogen, C ⁇ -C 4 -alkyl, C 4 haloalkyl, C ⁇ -C 4 alkoxy or C ⁇ -C 4 - Halogenalkoxy can wear, or
- Pyrazolyl-C -C 4 alkyl, which on Pyrazolrmg one or more substituents selected from the group consisting of halogen, C -C -alkyl, C 4 haloalkyl, C ⁇ -C 4 alkoxy or C ⁇ -C 4 - can carry haloalkoxy,
- R p 3 is hydrogen or -CC 4 alkyl
- Y is a carbon atom
- R n 1 one to five identical or different radicals from the group: hydrogen, halogen, C ⁇ -Cg-alkyl, Ci-Cg-alkoxy,
- R p 3 is hydrogen or methyl
- the amidoximes of the formula III are obtained by reacting nitriles of the formula II with hydroxylamine or its salts in aqueous solution, preferably in water or water / alkanol mixtures, if appropriate in the presence of a base. These can then be alkylated to the precursors IV in a manner known per se, cyclopropyl methyl bromide or cyclopropyl methyl chloride preferably being used as the alkylating agent.
- the iodide or organic sulfonic acid residues are also suitable for activating the cyclopropylmethyl residue.
- the compounds of formula 1 can preferably be prepared according to the following scheme:
- amidoximes IV can then be acylated in a manner known per se with the corresponding acid derivatives V, preferably the corresponding acid chlorides or acid anhydrides, by heating in inert solvents (preferably to temperatures in the range from 20 to 100 ° C.).
- inert solvents are in particular hydrocarbons or ethers, particularly preferably aromatic hydrocarbons such as toluene or xylene, to name just two examples.
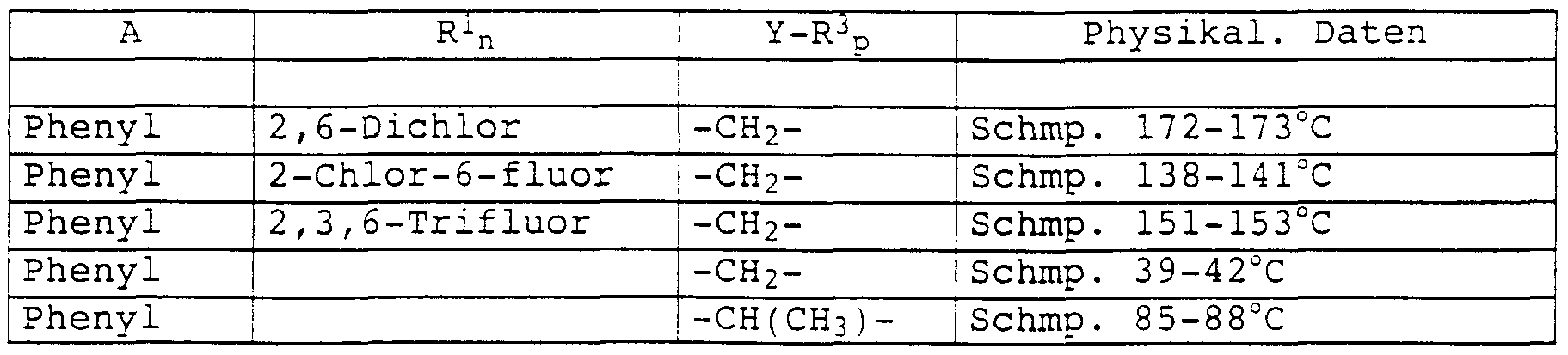
- Preferred amidoxime derivatives of the formula I are the compounds mentioned in Table 3, where R 2 is benzyl:
- the compounds I are distinguished by an excellent action Q against a broad spectrum of phytopathogenic fungi, in particular from the class of the Ascomycetes, Deuteromycetes, Phycomycetes and Basidiomycetes. Some of them are systemically effective and can therefore also be used as leaf and soil fungicides. 5 w «rt ⁇ * O g TJ H o XP ZI CD» 2J • no CC Tl TJ t? > g • v H 3 * H • s Cl HX ag ⁇ -3 ao 3 * O z
- magnesium sulfate magnesium oxide
- ground plastics fertilizers such as ammonium sulfate, ammonium phosphate, ammonium nitrate, ureas and vegetable products such as cereal flour, tree bark, wood and nutshell flour, cellulose powder or other solid carriers.
- V a mixture of 80 parts by weight, preferably a solid compound I according to the invention, 3 parts by weight of the sodium salt of di-iso-butylnaphthalene-2-sulfonic acid and 10 parts by weight of the sodium salt Lignm sulfonic acid from a sulfite waste liquor and 7 parts by weight of powdered silica gel; a fine boiling is obtained by finely distributing the mixture in water;
- a stable aqueous dispersion of 40 parts by weight of a compound I according to the invention, 10 parts by weight of the sodium salt of a phenolsulfonic acid-urea-formaldehyde condensate, 2 parts by weight of silica gel and 48 parts by weight of water Can be further diluted;
- IX a stable oily dispersion of 20 parts by weight of a compound I according to the invention, 2 parts by weight of the calcium salt of dodecylbenzenesulfonic acid, 8 parts by weight of fatty alcohol polyglycol ether, 20 parts by weight of the sodium salt of a phenolsulfonic acid-urea-formaldehyde Konoensates and 50 parts by weight of a paraffinic mineral oil.
- the new compounds are distinguished by excellent activity against a broad spectrum of phytopathogenic fungi, in particular from the classes of the Deuteromycetes, Ascomycetes, Phycomycetes and Basidiomycetes. Some of them are systemically effective and can be used as foliar and soil fungicides.
- the compounds are applied by treating the fungi or the seeds, plants, materials or the soil to be protected against fungal attack with a fungicidally active amount of the active compounds.
- the new compounds are particularly suitable for combating the following plant diseases:
- Erysiphe grammis (powdery mildew) on cereals, Erysiphe cichoracearum and Sphaerotheca fuligmea on pumpkin plants, Podosphaera leucot ⁇ cha on apples, Uncmula necator on vines, Puccmia species on cereals, Rhizoctonia species on cotton and turf, Ustilia sugar species maequalis (scab) on apples, Helmmthospor um species on vegetables Treide, Septo ⁇ a nodorum on wheat, Botrytis cinerea (gray mold) on strawberries, vines, ornamental plants and vegetables, Cercospora arachidicola on peanuts, Pseudocercosporella herpotrichoides on wheat, barley, Pyricula ⁇ a oryzae on rice, Phytophthora mfesticium Vertus and potatoes and tomatoes, Species on various plants, Plasmopara viticola on vines, Alternaria species on vegetables and fruits.
- the new compounds can also be used in material protection (wood protection), e.g. against Paecilomyces variotn.
- the fungicidal compositions generally contain between 0.1 and 95, preferably between 0.5 and 90% by weight of active ingredient.
- the application rates are between 0.025 and 2, preferably 0.1 to 1, kg of active ingredient per ha.
- active ingredient 0.001 to 50, preferably 0.01 to 10 g per kg of seed are generally required.
- agents according to the invention can also be present in the application form as fungicides together with other active ingredients, e.g. with herbicides, insecticides, growth regulators, fungicides or even with fertilizers.
- Sulfur, dithiocarbamates and their derivatives such as Fer ⁇ dimethyl- dithiocarbamate, Zmkdimethyldithiocarbamat, Zmkethylenbisdithio- carbamate, manganese ethylenebisdithiocarbamate, manganese ZmK-ethylenediamine mm bisdithiocarbamate, Tetramethylthiuramdisulfld, ammonia complex of Zmk- (N, N-ethylene-b ⁇ s-d ⁇ th ⁇ ocarbamat ), Ammonia complex of Zmk- (N, N '-propylene-bis-dithiocarbamate), zinc (N,' -propylene-bis-dithiocarbamate), N, N '-polypropylene-bis- (thio-carbamoyl) - disulfld;
- Nitrode ⁇ vate such as dinitro- (1-methylheptyl) phenylcrotonate, 2-sec. -Butyl-4, 6-dm ⁇ trophenyl-3, 3-dimethylacrylate, 2-sec. - Butyl-4, 6-dm ⁇ trophenyl- ⁇ so-propyl carbonate, 5-N ⁇ tro- ⁇ so-phthalic acid-di-iso-propyl ester; heterocyclic substances, such as 2-heptadecyl-2- ⁇ m ⁇ dazolm-acetate, 2, 4-d ⁇ chlor-6- (o-cnloranilmo) -s-t ⁇ azm, 0, O-diethyl-phthalimidophosphonothioate, 5-Ammo-l- [bis - (Dimethylamino) -phosphmyl] - 3-phenyl-l, 2,4-triazole, 2,3-diocyano-l, 4-dio-thioanthrachmon, 2-
- Strobilunne such as methyl-E-methoximino— [a— (o-tolyloxy) - o-tolyl] acetate, methyl-E-2— ⁇ 2— [6— (2-cyanophenoxy) pyri ⁇ imm-4-yl-oxy] phenyl ⁇ - 3 - ethoxyacrylate, methyl - E - methoximino - [a - (2, 5 - dimethylphenoxy) - o - tolyl] acetamide.
- Anilmo — pymids such as N— (4, 6 — dimethylpyrimidm — 2 — yl) anilm, N— [4 ⁇ methyl — 6— (1 — prop ⁇ nyl) py ⁇ m ⁇ d ⁇ n — 2 — yl] anilm, N— (4 — methyl— 6— cyclopropyl-pyrimidm-2-yl) anilm.
- Phenylpyrroles such as 4- (2,2-difluoro-1,3-benzodioxol-4-yl) -pyrrole-3-carbonitrile.
- Cinnamic acid amides such as 3-4 (4-chlorophenyl) -3- (3,4-dimethoxyphenyl) acrylic acid morpholide.
- aqueous active ingredient preparation which was prepared with a stock solution of 10% active ingredient, 63 ⁇ cyclohexanone and 27% emulsifier. 20 hours after the spray coating had dried on, the plants were inoculated with an aqueous spore suspension of the cucumber powdery mildew (Sphaerotheca fuligmea). The plants were then grown in a greenhouse at temperatures between 20 and 24 ° C. and 60 to 80% relative atmospheric moisture cultivated for 20 days. The extent of mildew development was then determined visually in% of the total leaf area.
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/089,148 US6881742B1 (en) | 1999-10-06 | 2000-10-05 | Benzyl amidoxime derivatives, intermediate products and method for their production and use as fungicides |
| EP00992195A EP1218339B1 (de) | 1999-10-06 | 2000-10-05 | Benzylaminodoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide |
| AT00992195T ATE251123T1 (de) | 1999-10-06 | 2000-10-05 | Benzylaminodoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide |
| CA002386513A CA2386513A1 (en) | 1999-10-06 | 2000-10-05 | Novel benzyl amidoxime derivatives, intermediate products and method for their production and use as fungicides |
| DK00992195T DK1218339T3 (da) | 1999-10-06 | 2000-10-05 | Benzylaminodoximderivater, mellemprodukter herfor og fremgangsmåde til fremstilling heraf og anvendelsen af disse som fungicider |
| JP2001528135A JP2003516933A (ja) | 1999-10-06 | 2000-10-05 | 新規ベンジルアミドキシム誘導体、その製造用の中間生成物及び方法、並びにその殺真菌剤としての使用 |
| AU28339/01A AU2833901A (en) | 1999-10-06 | 2000-10-05 | Novel benzyl amidoxime derivatives, intermediate products and method for their production and use as fungicides |
| DE50003942T DE50003942D1 (de) | 1999-10-06 | 2000-10-05 | Benzylaminodoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide |
| US11/061,470 US7101900B2 (en) | 1999-10-06 | 2005-02-22 | Benzyl amidoxime derivatives, intermediate products and method for their production and use as fungicides |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| DE19948266.7 | 1999-10-06 | ||
| DE19948266 | 1999-10-06 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10089148 A-371-Of-International | 2000-10-05 | ||
| US11/061,470 Division US7101900B2 (en) | 1999-10-06 | 2005-02-22 | Benzyl amidoxime derivatives, intermediate products and method for their production and use as fungicides |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2001025187A2 true WO2001025187A2 (de) | 2001-04-12 |
| WO2001025187A3 WO2001025187A3 (de) | 2001-11-01 |
Family
ID=7924784
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2000/009744 WO2001025187A2 (de) | 1999-10-06 | 2000-10-05 | Benzylamidoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide |
Country Status (11)
| Country | Link |
|---|---|
| US (2) | US6881742B1 (de) |
| EP (1) | EP1218339B1 (de) |
| JP (1) | JP2003516933A (de) |
| AT (1) | ATE251123T1 (de) |
| AU (1) | AU2833901A (de) |
| CA (1) | CA2386513A1 (de) |
| DE (1) | DE50003942D1 (de) |
| DK (1) | DK1218339T3 (de) |
| ES (1) | ES2210032T3 (de) |
| TW (1) | TWI229662B (de) |
| WO (1) | WO2001025187A2 (de) |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE540409C (de) * | 1927-02-12 | 1931-12-17 | Chemische Fabriken | Verfahren zur Herstellung von Tetrazolen |
| GB876079A (en) * | 1960-05-10 | 1961-08-30 | Wassermann S P A Soc It Per Sp | Halogen-substituted amidoximes and manufacturing process |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB285080A (en) | 1927-02-11 | 1929-04-18 | Albert Boehringer | Processes for preparing tetrazoles |
| AR205111A1 (es) * | 1974-03-11 | 1976-04-05 | Dow Chemical Co | Un procedimiento para producir para - toluensulfonato de 2-(4-bromofenil) acetamidina |
| US3991210A (en) | 1974-03-11 | 1976-11-09 | The Dow Chemical Company | Acetamidine urinary antiseptics |
| US5232940A (en) * | 1985-12-20 | 1993-08-03 | Hatton Leslie R | Derivatives of N-phenylpyrazoles |
| JPH1095771A (ja) * | 1996-09-19 | 1998-04-14 | Nippon Soda Co Ltd | アミドオキシム誘導体、その製造法及び農園芸用殺菌剤 |
-
2000
- 2000-10-05 TW TW089120773A patent/TWI229662B/zh not_active IP Right Cessation
- 2000-10-05 EP EP00992195A patent/EP1218339B1/de not_active Expired - Lifetime
- 2000-10-05 JP JP2001528135A patent/JP2003516933A/ja not_active Withdrawn
- 2000-10-05 DK DK00992195T patent/DK1218339T3/da active
- 2000-10-05 AT AT00992195T patent/ATE251123T1/de not_active IP Right Cessation
- 2000-10-05 WO PCT/EP2000/009744 patent/WO2001025187A2/de active IP Right Grant
- 2000-10-05 AU AU28339/01A patent/AU2833901A/en not_active Abandoned
- 2000-10-05 US US10/089,148 patent/US6881742B1/en not_active Expired - Fee Related
- 2000-10-05 ES ES00992195T patent/ES2210032T3/es not_active Expired - Lifetime
- 2000-10-05 CA CA002386513A patent/CA2386513A1/en not_active Abandoned
- 2000-10-05 DE DE50003942T patent/DE50003942D1/de not_active Expired - Fee Related
-
2005
- 2005-02-22 US US11/061,470 patent/US7101900B2/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE540409C (de) * | 1927-02-12 | 1931-12-17 | Chemische Fabriken | Verfahren zur Herstellung von Tetrazolen |
| GB876079A (en) * | 1960-05-10 | 1961-08-30 | Wassermann S P A Soc It Per Sp | Halogen-substituted amidoximes and manufacturing process |
Non-Patent Citations (4)
| Title |
|---|
| BOUALEM OUSSAID ET AL.: "Improved synthesis of oxadiazoles under microwave irradation" SYNTHETIC COMMUNICATIONS., Bd. 25, Nr. 10, 1995, Seiten 1451-1459, XP000926265 MARCEL DEKKER, INC., BASEL., CH ISSN: 0039-7911 * |
| CHEMICAL ABSTRACTS, vol. 62, no. 10, 10. Mai 1965 (1965-05-10) Columbus, Ohio, US; abstract no. 11732e, A. A. AROYAN ET AL.: "Synthesis of some amines amidoximes, and derivatives of guanidine" Spalte 11732; XP002164076 & IZV. AKAD. NAUK ARM. SSR, KHIM. NAUKI, Bd. 17, Nr. 5, 1964, Seiten 543-548, * |
| PATENT ABSTRACTS OF JAPAN vol. 1998, no. 69, 31. Juli 1998 (1998-07-31) & JP 10 095771 A (NIPPON SODA CO LTD), 14. April 1998 (1998-04-14) in der Anmeldung erwähnt -& JP 10 095771 A 14. April 1998 (1998-04-14) * |
| SEIGO SUZUE ET AL.: "Synthetic antimicrobials" CHEMICAL AND PHARMACEUTICAL BULLETIN., Bd. 21, 1973, Seiten 2146 -2160, XP000926253 PHARMACEUTICAL SOCIETY OF JAPAN. TOKYO., JP ISSN: 0009-2363 * |
Also Published As
| Publication number | Publication date |
|---|---|
| TWI229662B (en) | 2005-03-21 |
| DE50003942D1 (de) | 2003-11-06 |
| ES2210032T3 (es) | 2004-07-01 |
| WO2001025187A3 (de) | 2001-11-01 |
| JP2003516933A (ja) | 2003-05-20 |
| DK1218339T3 (da) | 2004-01-26 |
| US6881742B1 (en) | 2005-04-19 |
| ATE251123T1 (de) | 2003-10-15 |
| EP1218339A2 (de) | 2002-07-03 |
| EP1218339B1 (de) | 2003-10-01 |
| CA2386513A1 (en) | 2001-04-12 |
| US7101900B2 (en) | 2006-09-05 |
| AU2833901A (en) | 2001-05-10 |
| US20050187265A1 (en) | 2005-08-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1017670B1 (de) | Benzamidoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide | |
| EP0585751B1 (de) | N-Methylamide, Verfahren und Zwischenprodukte zu ihrer Herstellung sowie Verfahren zur Bekämpfung von Schädlingen | |
| DE19531813A1 (de) | Bisphenylamide | |
| EP0400417A1 (de) | Neue Oximether und diese enthaltende Fungizide | |
| EP0386561B1 (de) | Substituierte Oximether und Fungizide, die diese Verbindungen enthalten | |
| WO1999063821A1 (de) | Verwendung von 2-(n-phenylamino)pyrimidinen als fungizide sowie neue 2-(n-phenylamino)pyrimidine | |
| DE3827361A1 (de) | Oximether, verfahren zu ihrer herstellung und diese verbindungen enthaltende fungizide | |
| EP0159584A2 (de) | N-(Azolylcarbamoyl)-hydroxylamine und diese enthaltende Fungizide | |
| DE3923093A1 (de) | Neue 3-methoximinopropionsaeureester und diese enthaltende fungizide | |
| WO2001030154A2 (de) | Pyrazole enthaltende agrochemische zusammensetzungen und ihre verwendung als fungizide flanzenschutzmittel | |
| EP0582902B1 (de) | Benzylderivate und diese enthaltende Schädlingsbekämpfungsmittel | |
| EP0162246B1 (de) | N-(Arylpropyl)-azolylharnstoffe und diese enthaltende Fungizide | |
| EP0420091B1 (de) | Neue Anilinderivate und diese enthaltende Fungizide | |
| WO2001025187A2 (de) | Benzylamidoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide | |
| EP0438726B1 (de) | Ungesättigte Cyclohexylessigsäurederivate und diese enthaltende Pflanzenschutzmittel | |
| EP0984686A1 (de) | Verfahren zur bekämpfung von schadpilzen | |
| EP1133229B1 (de) | Fungizide mittel enthaltend als wirkstoffe pyrrolidone | |
| EP1017671A1 (de) | Neue benzamidoxim-derivate, zwischenprodukte und verfahren zu deren herstellung und deren verwendung als fungizide | |
| DE3906160A1 (de) | Ortho-substituierte 1-naphthylether und diese enthaltende fungizide | |
| EP1107946B1 (de) | Cycloalkylcarbonsäureamide, deren herstellung und verwendung als fungizide im agrarbereich | |
| EP0337211A1 (de) | Ortho-substituierte Carbonsäurebenzylester und diese enthaltende Fungizide | |
| WO2001019782A1 (de) | Cyclopropancarbonsäureamide, deren herstellung und verwendung | |
| DE3506814A1 (de) | 5-isoxazolcarbonsaeureamide, ihre herstellung und verwendung als fungizide | |
| DE19857273A1 (de) | Fungizide Mittel enthaltend als Wirkstoffe 2-Oxa-1,3,6,7-tetraaza-cyclopenta[alpha]naphthalin-Derivate und deren Verwendung bei der Behandlung von Pflanzen | |
| DD284797A5 (de) | Fungizide mittel |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AK | Designated states |
Kind code of ref document: A2 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A2 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| DFPE | Request for preliminary examination filed prior to expiration of 19th month from priority date (pct application filed before 20040101) | ||
| AK | Designated states |
Kind code of ref document: A3 Designated state(s): AE AG AL AM AT AU AZ BA BB BG BR BY BZ CA CH CN CR CU CZ DE DK DM DZ EE ES FI GB GD GE GH GM HR HU ID IL IN IS JP KE KG KP KR KZ LC LK LR LS LT LU LV MA MD MG MK MN MW MX MZ NO NZ PL PT RO RU SD SE SG SI SK SL TJ TM TR TT TZ UA UG US UZ VN YU ZA ZW |
|
| AL | Designated countries for regional patents |
Kind code of ref document: A3 Designated state(s): GH GM KE LS MW MZ SD SL SZ TZ UG ZW AM AZ BY KG KZ MD RU TJ TM AT BE CH CY DE DK ES FI FR GB GR IE IT LU MC NL PT SE BF BJ CF CG CI CM GA GN GW ML MR NE SN TD TG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 10089148 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 2001 528135 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2000992195 Country of ref document: EP Ref document number: 2386513 Country of ref document: CA |
|
| WWP | Wipo information: published in national office |
Ref document number: 2000992195 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: DE Ref legal event code: 8642 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2000992195 Country of ref document: EP |